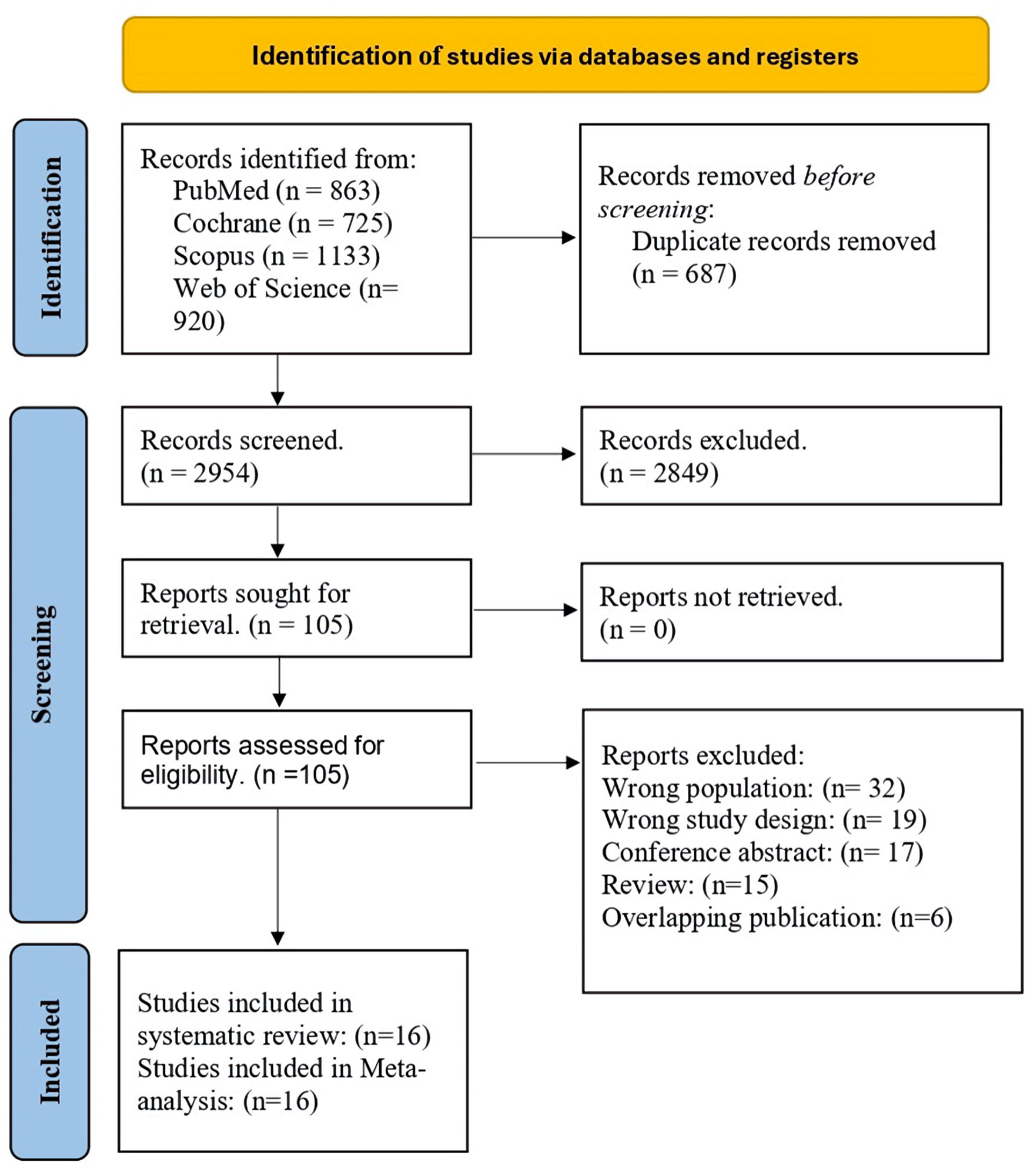
↓ Figure 1. PRISMA flow diagram. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
| Journal of Clinical Medicine Research, ISSN 1918-3003 print, 1918-3011 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Med Res and Elmer Press Inc |
| Journal website https://jocmr.elmerjournals.com |
Original Article
Volume 18, Number 3, March 2026, pages 177-195
Restrictive Versus Liberal Fluid Strategy for Initial Resuscitation in Sepsis and Septic Shock: A Systematic Review and Meta Analysis
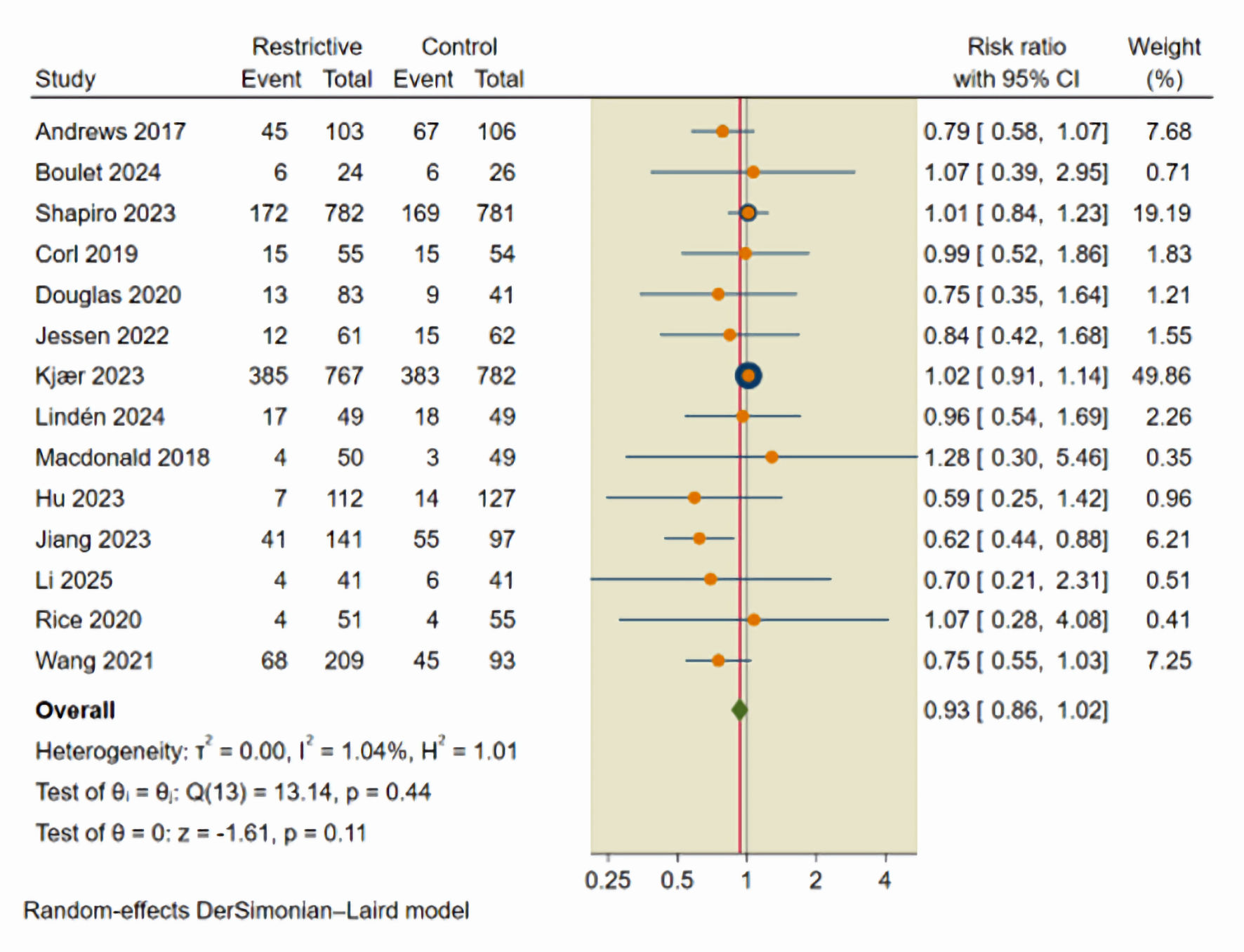
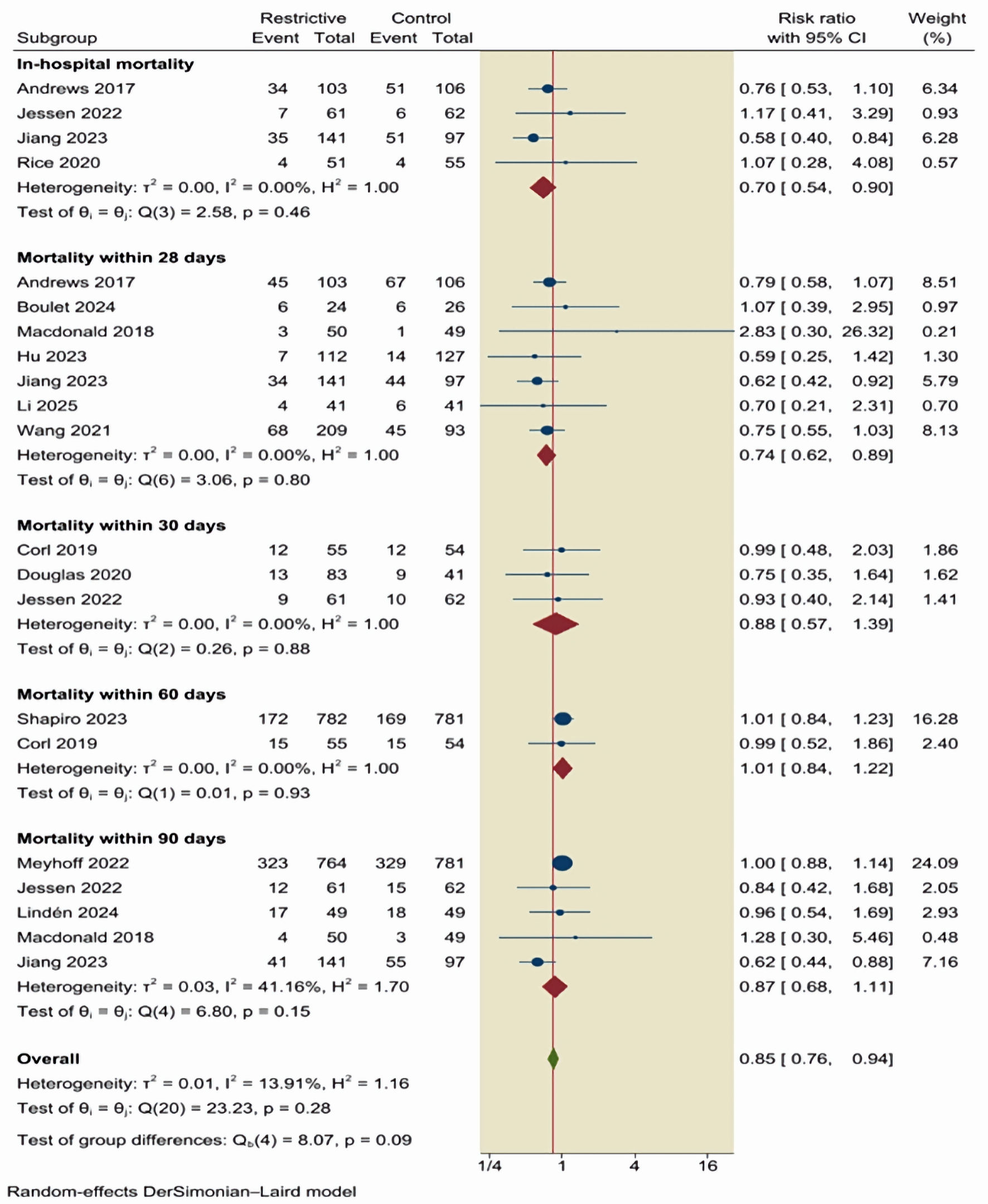
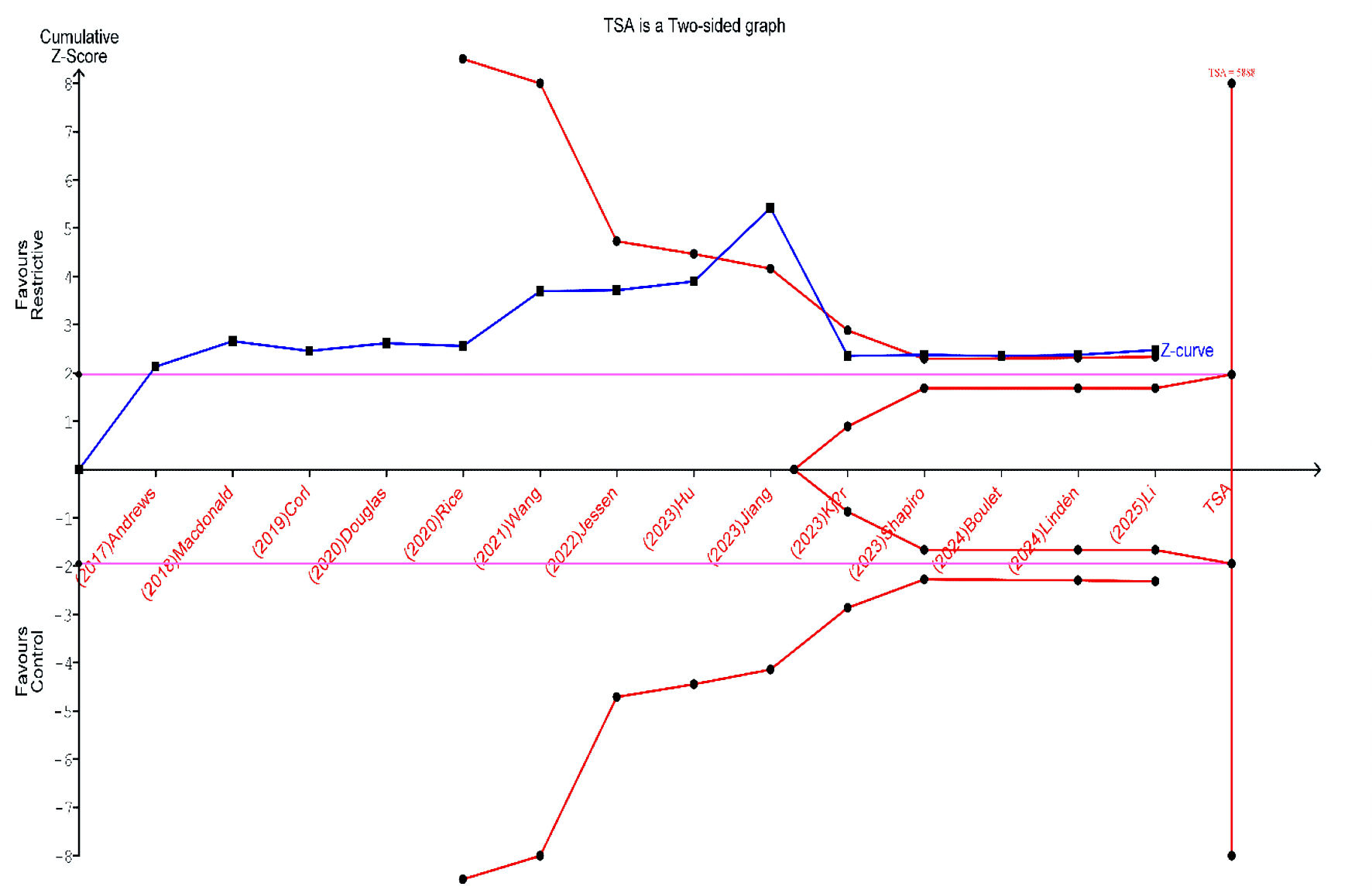
Figures

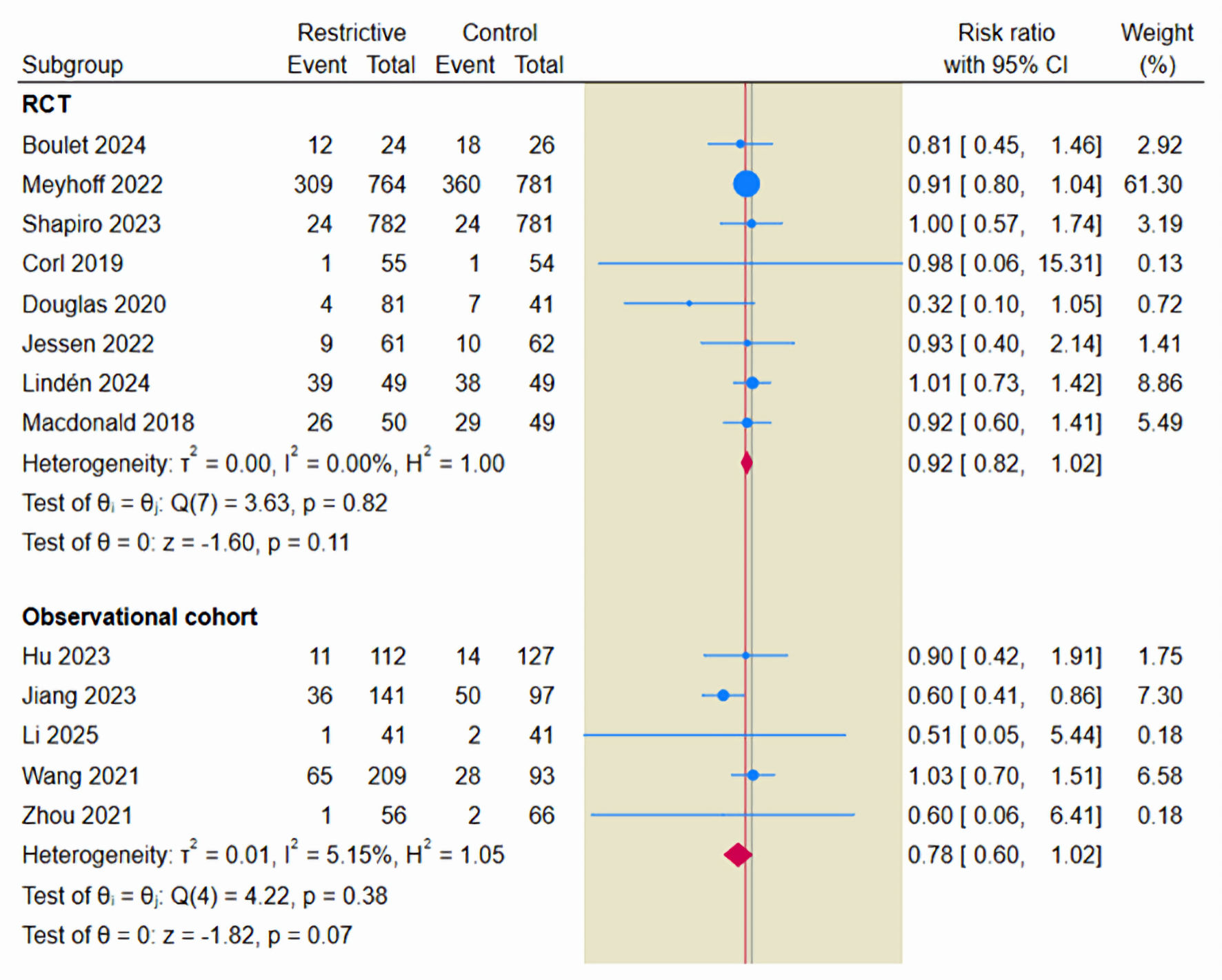
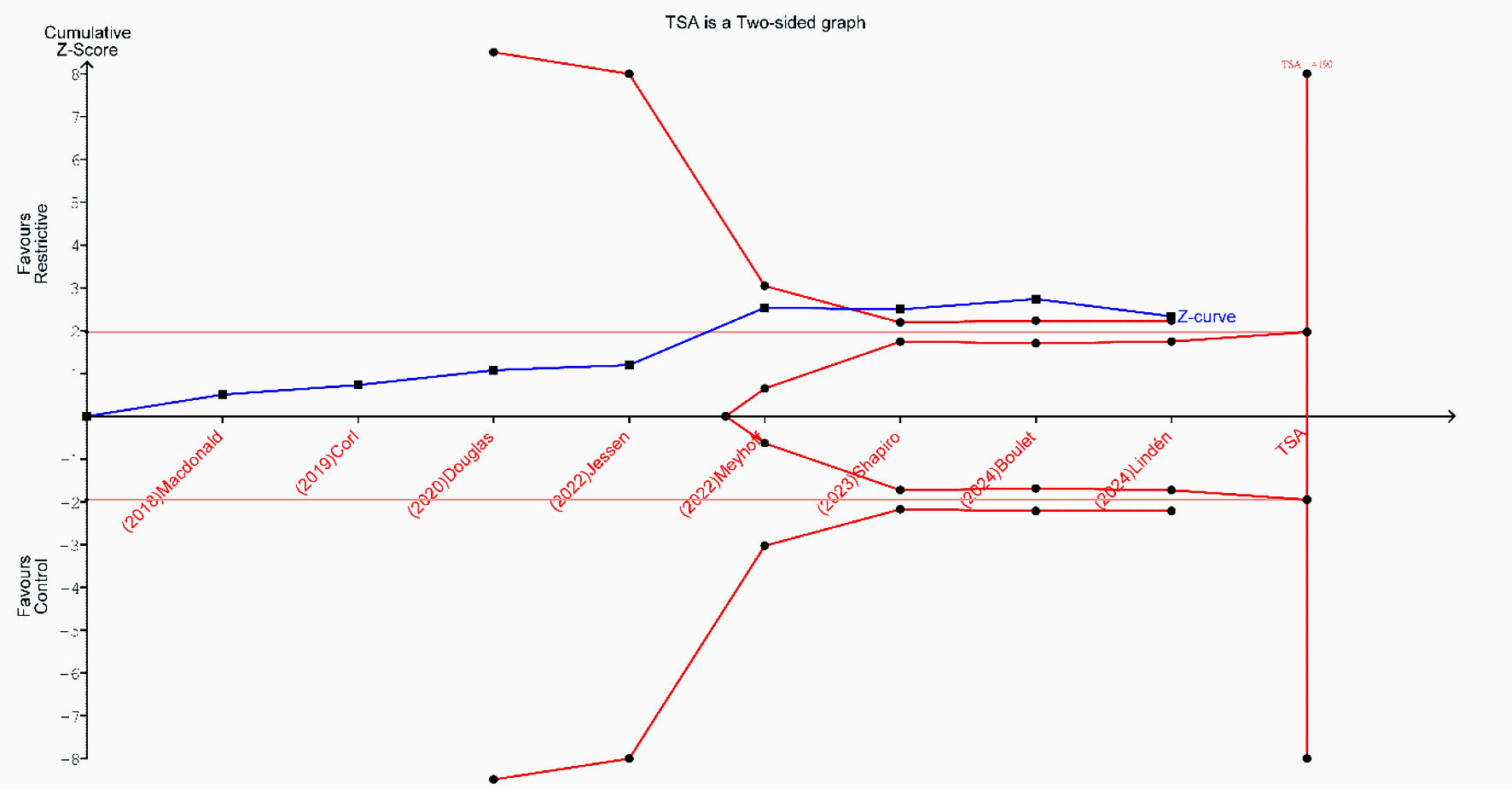
Tables
| Study ID | Groups | Patient demographics | Hemodynamic parameters, mean (SD) | Focus of infection, n (%) | Comorbidities, n (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Age (years), mean (SD) | Weight (kg), mean (SD) | Male, n (%) | SOFA score | Lactate, mmol/L | Creatinine (µmol/L) | Pulmonary | Urinary tract | Skin or soft tissue | Diabetes | Hypertension | CKD | ||
| Data are presented as: n (%) or mean (SD). NA: not available; SOFA: Sequential Organ Failure Assessment; CKD: chronic kidney disease; SD: standard deviation. | |||||||||||||
| Andrews et al, 2017 [33] | Restrictive fluid | 35.8 (11.9) | NA | 55 (53.4) | NA | 4.5 (3.3) | 141.5 (119.3) | NA | 2 (1.9) | NA | NA | NA | NA |
| Standard fluid | 37.5 (12.9) | NA | 62 (58.5) | NA | 5.4 (4.4) | 141.5 (88.4) | NA | 2 (1.9) | NA | NA | NA | NA | |
| Boulet et al, 2024 [21] | Restrictive fluid | 69.5 (8.8) | 67.7 (14.8) | 12 (50) | NA | 2.4 (1.8) | 98.5 (52) | 12 (50) | 1 (4.2) | 1 (4.2) | 8 (33.3) | 14 (58.3) | 1 (4.2) |
| Standard fluid | 67.0 (11.5) | 73.4 (16.6) | 13 (54.2) | NA | 2.2 (1.1) | 158.8 (47.6) | 11 (45.8) | 5 (16.7) | 1 (4.2) | 5 (20.8) | 11 (45.8) | 2 (8.3) | |
| Meyhoff et al, 2022 [13] | Restrictive fluid | 70 (11.4) | 78 (17) | 452 (59.9) | NA | 4.1 (2.4) | 150.2 (84.8) | 209 (27.7) | 119 (15.8) | 62 (8.2) | NA | 346 (45.8) | 9 (1.2) |
| Standard fluid | 69 (12.6) | 78.6 (17.8) | 452 (58.2) | NA | 4.2 (2.4) | 153.8 (91) | 206 (26.5) | 133 (17.1) | 64 (8.2) | NA | 360 (46.4) | 12 (1.5) | |
| Shapiro et al, 2023 [14] | Restrictive fluid | 59.1 (16) | NA | 411 (52.6) | 3.4 (2.8) | 2.9 (2.5) | 159.12 (50.2) | 217 (27.7) | 148 (18.9) | 97 (12.4) | 222 (28.6) | NA | 33 (4.2) |
| Standard fluid | 59.9 (15.9) | NA | 415 (53.1) | 3.5 (2.7) | 2.9±2.4 | 167.9 (167.9) | 205 (26.2) | 172 (22.0) | 82 (10.5) | 224 (29.0) | NA | 40 (5.2) | |
| Corl et al, 2019 [22] | Restrictive fluid | 71 (16.7) | 87.7 (21.3) | 24 (43.6) | 8.7 (3) | 2.3 (1.7) | NA | 13 (23.6) | 15 (27.3) | 8 (14.6) | 23 (41.8) | NA | 7 (12.7) |
| Standard fluid | 69.5 (20.5) | 80.1 (17) | 26 (48.2) | 9.1 (3.8) | 3.2 (2.8) | NA | 17 (31.5) | 14 (25.9) | 2 (3.7) | 18 (33.3) | NA | 1 (1.9) | |
| Douglas et al, 2020 [34] | Restrictive fluid | 61.8 (16.9) | 73.7 (18.7) | 32 (38.6) | NA | 3.6 (3.2) | NA | NA | NA | NA | 29 (34.9) | 42 (50.6) | NA |
| Standard fluid | 62.7 (15) | 73.6 (18.5) | 28 (68.3) | NA | 3.8 (3.6) | NA | NA | NA | NA | 11 (26.8) | 25 (61) | NA | |
| Jessen et al, 2022 [23] | Restrictive fluid | 75.7 (13.7) | 77 (21.2) | 37 (61) | 2.7 (0.75) | 1.4 (0.6) | 98 (53.9) | 45 (74) | 9 (15) | 3 (5) | 11 (18) | NA | 5 (8) |
| Standard fluid | 75.7 (11.3) | 78.7 (15.9) | 34 (55) | 2.7 (0.75) | 1.5 (0.8) | 96 (50.8) | 43 (69) | 12 (19) | 1 (2) | 9 (15) | NA | 9 (15) | |
| Kjaer et al, 2023 [24] | Restrictive fluid | 70 (11.4) | 78 (17) | 452 (59.9) | NA | 4.1 (2.4) | 150.2 (84.8) | 209 (27.7) | 119 (15.8) | 62 (8.2) | NA | 346 (45.8) | 9 (1.2) |
| Standard fluid | 69 (12.6) | 78.6 (17.8) | 452 (58.2) | NA | 4.2 (2.4) | 153.8 (91) | 206 (26.5) | 133 (17.1) | 64 (8.2) | NA | 360 (46.4) | 12 (1.5) | |
| Linden et al, 2024 [25] | Restrictive fluid | 71.4 (16) | 82.7 (20.7) | 26 (57) | 10.4 (3.8) | 4.2 (3.1) | 172.4 (101) | 13 (28) | 12 (26) | 5 (11) | NA | NA | NA |
| Standard fluid | 68.7 (13.7) | 78.7 (22.1) | 30 (61) | 9.4 (3.8) | 4 (2.7) | 172 (148.16) | 17 (35) | 7 (14) | 3 (6) | NA | NA | NA | |
| Macdonald et al, 2018 [26] | Restrictive fluid | 65.4 (19.8) | 78 (16.7) | 31 (62) | 5.7 (4.5) | 2.1 (1.8) | 113.7 (64.8) | 14 (28) | 16 (32) | 6 (12) | NA | NA | NA |
| Standard fluid | 62.4 (23.6) | 75.4 (19.8) | 30 (61) | 5.4 (2.2) | 1.8 (1) | 126.7 (68.7) | 20 (41) | 9 (18) | 6 (12) | NA | NA | NA | |
| Hu et al, 2023 [27] | Restrictive fluid | 66.28 (16.66) | NA | 67 (59.82) | 6.02 (2.97) | NA | 147.29 (185.58) | NA | NA | NA | NA | NA | NA |
| Standard fluid | 67.25 (19.41) | NA | 77 (60.63) | 7.12 (2.99) | NA | 94.03 (65.66) | NA | NA | NA | NA | NA | NA | |
| Jiang et al, 2023 [28] | Restrictive fluid | 64.8 (12.7) | NA | 96 (68.1) | 8.5 (3.4) | 3.1 (2.1) | NA | 53 (37.6) | 18 (12.8) | 8 (5.7) | 29 (20.6) | 57 (40.4) | NA |
| Standard fluid | 64.7 (13.8) | NA | 61 (62.9) | 9.3 (3.2) | 3.9 (2.7) | NA | 31 (32.0) | 5 (5.2) | 5 (5.2) | 30 (30.9) | 43 (44.3) | NA | |
| Li et al, 2025 [29] | Restrictive fluid | 56.24 (9.79) | NA | 24 (58.5) | NA | NA | NA | 16 (39) | NA | NA | 8 (19.5) | 19 (46.3) | 2 (4.9) |
| Standard fluid | 56.56 (12.07) | NA | 26 (63.4) | NA | NA | NA | 15 (36.6) | NA | NA | NA | 22 (53.7) | 3 (7.3) | |
| Rice et al, 2020 [30] | Restrictive fluid | 71.2 (11.9) | 88.3 (27.1) | 28 (54.9) | 7 (4) | NA | NA | NA | NA | NA | NA | NA | NA |
| Standard fluid | 72.5 (10.6) | 84.9 (23.1) | 27 (49.1) | 6 (4) | NA | NA | NA | NA | NA | NA | NA | NA | |
| Zhou et al, 2021 [32] | Restrictive fluid | 40.19 (3.22) | 60.19 (2.39) | 30 (53.6) | NA | NA | NA | 20 (35.7) | NA | 14 (25) | NA | NA | NA |
| Standard fluid | 39.98 (3.35) | 60.28 (2.31) | 40 (60.6) | NA | NA | NA | 23 (34.8) | NA | 13 (19.7) | NA | NA | NA | |
| Wang et al, 2021 [31] | Restrictive fluid | 68.4 (15) | NA | 78 (37) | 12.3 (2.9) | 4.8 (2.7) | NA | 141 (67.4) | 7 (3.3) | NA | 36 (17.3) | 48 (22.9) | 8 (3.8) |
| Standard-fluid | 69.6 (13) | NA | 28 (30) | 12.3 (3.7) | 4.9 (2.5) | NA | 58 (62.4) | 4 (4.3) | NA | 17 (18.3) | 33 (35.5) | 3 (3.2) | |
| Study | Study design | Country | Time frame | Follow-up (days) | Sample size | Inclusion criteria | Primary outcomes | ||
|---|---|---|---|---|---|---|---|---|---|
| Total | Restrictive strategy | Standard care | |||||||
| LOS: length of stay; SIRS: Systemic Inflammatory Response Syndrome; SBP: systolic blood pressure; BP: blood pressure; ED: emergency department; ICU: intensive care unit; IV: intravenous; Sepsis-3: Third International Consensus Definitions for Sepsis and Septic Shock; SCCM: Society of Critical Care Medicine; ESICM: European Society of Intensive Care Medicine; APACHE II: Acute Physiology and Chronic Health Evaluation II; SOFA: Sequential Organ Failure Assessment; ARDS: acute respiratory distress syndrome; MODS: multiple organ dysfunction syndrome; HR: heart rate; MAP: mean arterial pressure; CVP: central venous pressure; CO: cardiac output; SV: stroke volume; LVEF: left ventricular ejection fraction; cTnI: cardiac troponin I; NT-proBNP: N-terminal pro-B-type brain natriuretic peptide; CRP: C-reactive protein. | |||||||||
| Andrews et al, 2017 [33] | Parallel-group, non-blinded, randomized clinical trial (RCT) | Zambia | October 22, 2012, to November 11, 2013 | 28 days | 209 | 103 | 106 | Adults ≥ 18 years with sepsis (suspected infection + ≥ 2 SIRS criteria) and hypotension (SBP ≤ 90 mm Hg or MAP ≤ 65 mm Hg), enrolled within 4 h of the first eligible BP and within 24 h of ED registration | In-hospital mortality |
| Boulet et al, 2024 [21] | Single-blind, randomized, parallel, controlled pilot trial | France | September 20, 2021, to February 25, 2023 | 28 days | 50 | 24 | 26 | Adults 18–85 years, admitted to ICU with septic shock and within the first 24 h of vasopressor infusion | Cumulative fluid balance (mL/kg of admission body weight) in the first 5 days |
| Meyhoff et al, 2022 [13] | Open label randomized clinical trial | 31 ICUs across Denmark, Norway, Sweden, Switzerland, Italy, Czech Republic, UK, and Belgium | November 27, 2018, to November 16, 2021 | 90 days | 1,545 | 764 | 781 | Adults ≥ 18 y in ICU with septic shock, received ≥ 1 L IV fluid in prior 24 h, onset of shock ≤ 12 h before screening | Death from any cause within 90 days |
| Shapiro et al, 2023 [14] | Randomized, unblinded superiority trial | USA | March 7, 2018, to January 31, 2022 | 90 days | 1,563 | 782 | 781 | Adults ≥ 18 years with suspected or confirmed infection who had sepsis-induced hypotension, defined in the trial as systolic blood pressure < 100 mm Hg after administration of ≥ 1,000 mL IV fluid | Death from any cause before discharge home by day 90 |
| Corl et al, 2019 [22] | Prospective, randomized controlled trial | USA | November 2016 to February 2018 | 60 days | 109 | 55 | 54 | Adults admitted from the ED to the ICU with suspected severe sepsis or septic shock. After receiving a 1-L fluid bolus, they were included if they had persistent hypotension (MAP < 65 mm Hg) or lactate ≥ 4 mmol/L. | 30-day all-cause mortality |
| Douglas et al, 2020 [34] | Prospective, multicenter, randomized, unblinded clinical trial | USA and UK | October 2016 to February 2019 | 30 days | 124 | 83 | 41 | Adults presenting to ED with sepsis or septic shock (paper used SIRS-based sepsis criteria: ≥ 2 SIRS criteria + suspected/documented infection) and anticipated ICU admission; refractory hypotension defined as MAP ≤ 65 mm Hg after receiving ≥ 1 L and < 3 L of fluid; enrolment within 24 h of hospital arrival | Difference in positive fluid balance at 72 h or ICU discharge |
| Jessen et al, 2022 [23] | Randomized, parallel-group, open-label feasibility trial | Denmark | November 3, 2021, to December 18, 2021 | 90 days | 123 | 61 | 62 | Adult age ≥ 18 years; sepsis defined by suspected infection by treating clinician, blood cultures drawn, IV antibiotics given or planned, infection-related increase in SOFA ≥ 2; expected hospital stay > 24 h | Total volume of IV crystalloid fluids administered during the first 24 h |
| Kjaer et al, 2023 [24] | Long-term follow of the CLASSIC trial | 31 ICUs across Denmark, Norway, Sweden, Switzerland, Italy, Czech Republic, UK and Belgium | November 27, 2018, to November 16, 2021 | 365 days | 1,549 | 767 | 782 | Adults ≥18 years in ICU with septic shock, received ≥ 1 L IV fluid in prior 24 h, onset of shock ≤ 12 h before screening | Death from any cause within 1 year |
| Linden et al, 2024 [25] | Multicenter, parallel group, randomized feasibility trial | Sweden | March 7 and September 13, 2022 | 90 days | 98 | 49 | 49 | Adult patients (≥ 18 years of age) with septic shock (suspected/confirmed infection, plasma lactate > 2 mmol/L, and infusion of vasopressor to maintain MAP > 65 mm Hg after adequate fluid resuscitation) within 12 h of admission to the ICU and ongoing vasopressor therapy at the time of inclusion were eligible for inclusion | Total volume of fluid administered within 3 days |
| Macdonald et al, 2018 [26] | Multicenter, prospective, randomized, open-label clinical trial | Australia | October 2016 to March 2018 | 90 days | 99 | 50 | 49 | Adults with suspected infection requiring IV antibiotics and hypotension, defined as SBP < 100 mm Hg despite at least 1,000 mL isotonic crystalloid given within ≤ 1 h before randomization (SBP threshold was originally < 90 mm Hg, but changed to < 100 mm Hg early due to slow recruitment | Cumulative total IV fluid volume administered at 6 h post-randomization |
| Hu et al, 2023 [27] | Retrospective cohort study | China | January to September 2020 | 28 days | 239 | 112 | 127 | Adults ≥ 18 years, first onset, meeting the 2012 international guidelines diagnostic criteria for severe sepsis/septic shock, and no treatment contraindications | ICU stay time, ventilator use time, 28-day mortality, hemodynamic indices, APACHE II and SOFA scores, lactate clearance, complications (ARDS, acute renal failure, MODS), and myocardial injury markers |
| Jiang et al, 2023 [28] | Retrospective cohort study | China | January 2019 to December 2022 | 90 days | 238 | 141 | 97 | Age ≥ 18 years, diagnosed with septic shock based on clinical, microbiologic, radiologic, or intraoperative findings, and required IV fluid resuscitation during the acute critical phase | In-hospital mortality |
| Li et al, 2025 [29] | Retrospective cohort study | China | January 2021 to December 2023 | 28 dayss | 82 | 41 | 41 | Septic shock per Sepsis-3 SCCM/ESICM definition. Age 18–75 years; normal immune function; APACHE II ≥ 12; crystalloid fluid resuscitation required. | 28-day all-cause mortality, fluid infusion volume, and ICU LOS |
| Rice et al, 2020 [30] | Retrospective cohort study | USA | October 1, 2016, to July 31, 2019 | 30 days | 106 | 51 | 55 | Adults ≥ 18 years with severe sepsis or septic shock, documented chronic kidney disease (CKD), and ≥ 1 episode of hypotension within 6 h of ED presentation | Total hospital LOS |
| Zhou et al, 2021 [32] | Retrospective cohort study | China | January 2019 to January 2020 | 64 days | 122 | 56 | 66 | Clinical diagnosis of sepsis, complete medical records, ethics-approved management, and patient or family consent | Hemodynamic changes (HR, MAP, CVP), cardiac function (CO, SV, LVEF), biomarkers (cTnI, NT-proBNP, CRP), survival rate up to 64 days, and complication incidence |
| Wang et al, 2021 [31] | Prospective cohort study | China | May 8, 2018, to June 15, 2021 | 28 days | 302 | 209 | 93 | Age ≥ 18 present with septic shock: infection + SOFA increase ≥ 2, and persistent hypotension needing vasopressors to maintain MAP > 65 mm Hg, and lactate > 2 mmol/L despite fluids | 28-day mortality |