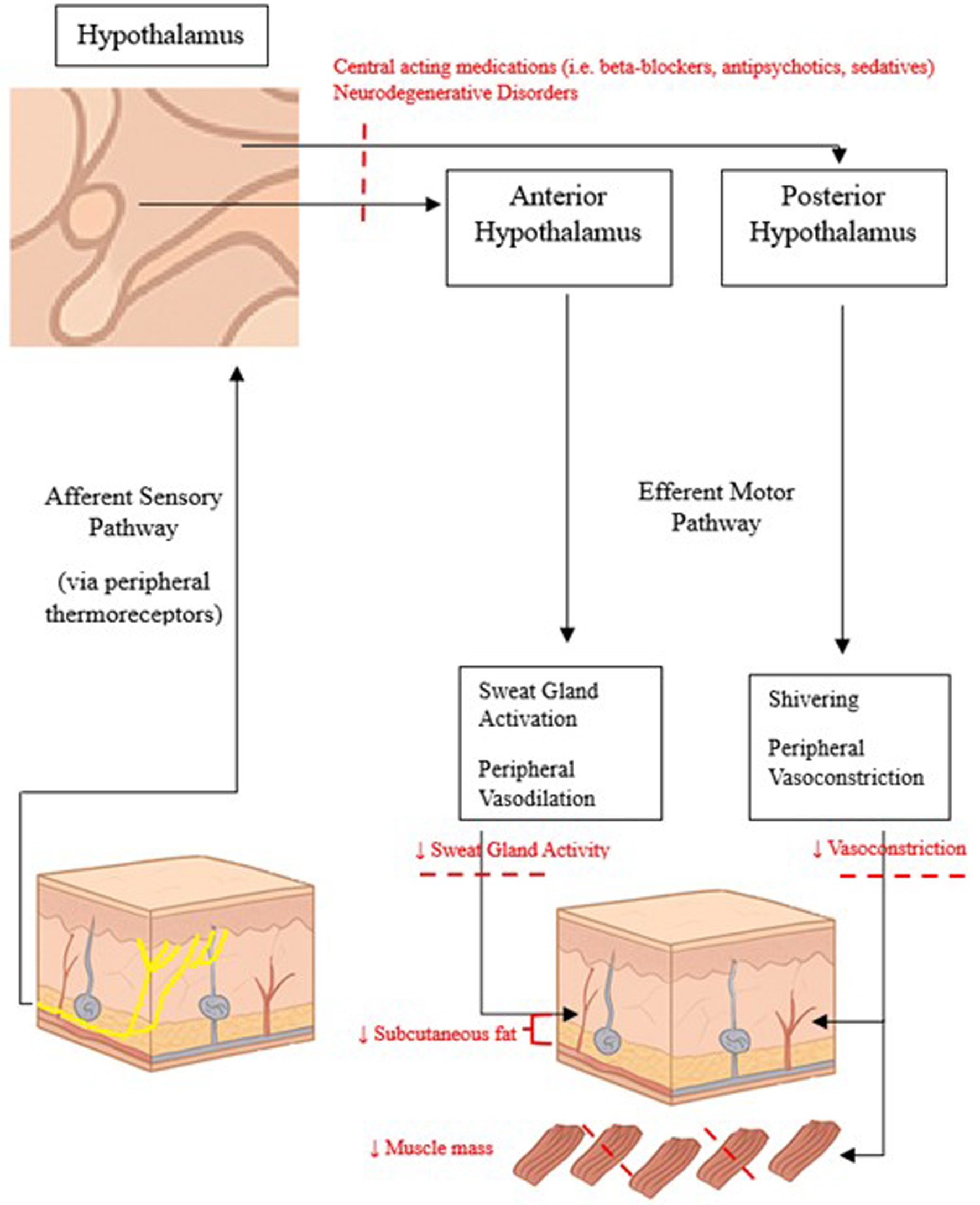
↓ Figure 1. Normal thermoregulatory pathways and changes associated with aging.
| Journal of Clinical Medicine Research, ISSN 1918-3003 print, 1918-3011 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Med Res and Elmer Press Inc |
| Journal website https://jocmr.elmerjournals.com |
Review
Volume 18, Number 4, April 2026, pages 219-233
Approach to Low Body Temperature or Mild Hypothermia in the Geriatric Population: A Narrative Review
Figure

Tables
| Accuracy rank | Measurement site | Instrument/technique | Advantages | Disadvantages | References |
|---|---|---|---|---|---|
| Best | Pulmonary artery | Swan-Ganz catheter | 1. Gold standard for measuring core body temperature as measures directly from central circulation. 2. Highly precise and repeatable. | 1. Highly invasive, requires intravascular access. 2. Limited to intensive care unit (ICU) and operative settings. | Hymczak et al, 2021 [35] |
| Distal esophagus | Long esophageal thermistor | 1. Strongly correlated with pulmonary artery temperature. 2. Changes rapidly in response to core temperature changes. 3. Preferred in intubated patients. | 1. Lags during rapid rewarming/cooling as seen in cardiac surgery. 2. Not useful for awake patients. | Lefrant et al, 2003 [36]; Misra et al, 2023 [37]; Sessler, 2016 [34] | |
| Good | Nasopharynx | Nasopharyngeal temperature probe | 1. Anatomically close to internal carotid artery. 2. Reflects core temperature during anesthesia. 3. Widely available. | 1. Difficult to position correctly (highly practitioner dependent). | Lee et al, 2014 [40]; Lim et al, 2016 [39] |
| Urinary bladder | Temperature-sensing urinary catheter | 1. Offers continuous temperature monitoring. 2. Reliable and precise during steady states. | 1. Temperatures lag behind rapid core temperature shifts (as seen in cardiopulmonary bypass). 2. Limited to patients with urinary catheters. | Fallis, 2002 [32] | |
| Rectum | Rectal probe (≥ 15 cm insertion) | 1. Easy to use. 2. Correlates well with intravascular temperatures under stable conditions. | 1. Delayed response during rapid temperature change. 2. Temperatures are affected by presence of feces or inflammation. 3. Risk of transmitting infection (e.g., VRE transmission). 4. Patient discomfort. | Robinson et al, 1998 [50]; Livornese et al, 1992 [51]; Niven et al, 2015 [48] | |
| Moderate | Tympanic membrane | Infrared or insulated thermistor | 1. Non-invasive. 2. Approximates brain core temperature (shares blood supply with hypothalamus). | 1. Accuracy reduced by cerumen, ear pathology, or poor placement technique. | Mah et al, 2021 [47]; Gasim et al, 2013 [46]; Sund-Levander & Grodzinsky, 2020 [31] |
| Poor | Temporal artery (forehead) | Infrared temporal scanner | 1. Contactless and thus low infection transmission risk. 2. Does not require patient to be awake. | 1. Highly influenced by ambient temperature, sweat, and poor technique. 2. Does not correlate well with true core temperature. | Chen et al, 2020 [43]; Hussain et al, 2021 [44]; Chen et al, 2021 [45] |
| Oral | Oral electronic thermometer | 1. Convenient, accessible. 2. Non-invasive and widely available. | 1. Inaccurate during thermal stress, recent oral intake, or after smoking. 2. Underestimates true core body temperature and possible fever. | Mazerolle et al, 2011 [41]; Betta et al, 1997 [42]; Ley et al, 2023 [19] | |
| Axilla | Axillary thermometer | 1. Widely available and easy to use. 2. Safe alternative when oral or rectal routes are contraindicated. | 1. Poorly correlates with core body temperature. 2. Strongly affected by ambient temperature and skin moisture. | Sund-Levander & Grozsinsky, 2020 [31]; Ley et al, 2023 [19] |
| Clinical correlate | Temperature range (°C) |
|---|---|
| Hypothermia | < 35.0 |
| Low body temperature | 35.0–36.0 |
| Normothermia | 36.1–37.2 |
| Low-grade fever | 37.3–37.9 |
| True fever | ≥ 38 |
| Comorbidity | Pathophysiologic link to low body temperature | Clinical consequences | Supporting references |
|---|---|---|---|
| Chronic kidney disease (CKD) | 1. Hypothermia causes decreased cardiac output and lowers glomerular filtration rate leading to pre-renal injury. 2. Endogenous cryogens and uremic toxins suppress normal thermoregulatory responses. 3. CKD-related malnutrition and anemia lead to impaired thermogenesis and reduced heat conservation. | 1. High prevalence of acute kidney injury (AKI) in hypothermic patients. 2. Hemodialysis patients have a lower baseline body temperature prior to dialysis. 3. Cold skin and cold intolerance in CKD-associated anemic patients, improving with erythropoietin administration. | Mallet, 2002 [61]; Kuriyama et al, 1999 [63]; Yamada et al, 2010 [62]; Noe et al, 2012 [64]; Kluger et al, 1981 [65]; Ash, 1991 [66]; Fishbane & Spinowitz, 2018 [67]; Brigham & Beard, 1996 [68]; Ludwig & Strasser, 2001 [69]; Grassi et al, 2003 [21]; Kenney & Munce, 2003 [16] |
| Hypertension | 1. Cold exposure leads to increased sympathetic nervous system activity and RAAS activation, which causes vasoconstriction and increased vascular resistance. | 1. Cold-induced blood pressure elevation. 2. Higher mortality in hypertensive elderly during winter months. | Goel et al, 2022 [70]; Hu et al, 2021 [71]; Wang et al, 2017 [72]; Greaney et al, 2017 [73]; Qi et al, 2024 [74] |
| Diabetes mellitus | 1. Autonomic and small fiber neuropathy leads to impaired shivering, vasoconstriction, and thermoregulation. 2. Hypoglycemia may precipitate or result from hypothermia. | 1. Increased cold-related morbidity and mortality. 2. Frequent hypoglycemic episodes in elderly patients during low ambient temperatures. | Cheshire, 2016 [77]; Tran et al, 2012 [78]; Lai et al, 2020 [79]; Song et al., 2021 [76]; Yang et al, 2016 [75]; Ali Imuran et al, 2018 [80]; American Diabetes Association Professional Practice Committee, 2025 [81] |
| Chronic obstructive pulmonary disease (COPD) | 1. Chronic hypoxemia and polyneuropathy impair autonomic thermoregulation. 2. Cold exposure worsens pulmonary function. | 1. Increased exacerbations and hospitalizations during cold weather. 2. Low body temperature correlates with disease severity. | Yang et al, 2024 [82]; Lam et al, 2018 [83]; Li et al, 2022 [84]; Aras et al, 2018 [86]; Chhabra & De, 2005 [87]; Arisoy et al, 2021 [85] |
| Malignancy | 1. Cold-induced sympathetic activation and β-adrenergic signaling promote carcinogenic pathways, angiogenesis, and metastasis formation. 2. Sarcopenia from malignancy leads to decreased insulation and heat retention. | 1. Increased cancer incidence, mortality, and treatment resistance in colder climates. 2. Increased susceptibility to hypothermia in sarcopenic patients. | Bandyopadhayaya et al, 2020 [88]; Mravec & Tibensky, 2020 [90]; Eng et al, 2015 [91]; Zhang et al, 2023 [92]; Obermeyer et al, 2017 [93]; Sørensen et al, 2005 [94] |
| Parkinson’s disease | 1. Central thermoregulatory dysfunction from α-synuclein deposits and Lewy bodies in hypothalamus. 2. Peripheral autonomic and sensory denervation. | 1. Impaired sweating, vasodilation, and shivering via normal autonomic regulatory pathways. 2. D2 receptor agonist mediated thermoregulatory changes. | Coon & Low, 2018 [95]; De Marinis et al, 1991 [97]; Doppler et al, 2014 [96]; Hama et al, 2009 [98] |
| Polypharmacy | 1. Impaired thermoregulatory mechanisms via anxiolytics, antipsychotics, antidepressants, and opioids. 2. Weakening compensatory response to low ambient temperatures (β-blockers, α1-adrenergic receptor agonists, and oral antihyperglycemics). | 1. Drug-induced hypothermia. 2. Impaired recognition of hypoglycemia. 2. Altered drug metabolism in mild hypothermia. | Van Marum et al, 2007 [29]; Zonnenberg et al, 2017 [101]; Nemmani et al, 2001 [104]; Liu et al, 2016 [102]; Vue & Setter, 2011 [110]; Lin et al, 1984 [112]; Tortorici et al, 2007 [113]; Ben-Uriah et al, 1981 [109] |
| Dementia/cognitive decline | 1. Low body temperature leads to tau hyperphosphorylation and amyloid accumulation. 2. Hypothalamic involvement in neurodegeneration. | 1. Worsening cognition. 2. Increased risk of conversion to dementia. 3. Low body temperature as a prodromal phase of dementia. | Vandal et al, 2016 [116]; Blessing et al, 2022 [117]; Van De Nes et al, 1998 [118]; Alagiakrishnan et al, 2023 [119]; Yamagiwa et al, 2025 [115]; Fischer et al, 2024 [120]; Wang et al, 2025 [121] |
| Frailty | 1. Low body temperature is associated with reduced metabolic rate, loss of muscle mass, and impaired physiologic reserve, all of which contribute to frailty. | 1. Lower body temperatures are associated with increased risk of frailty. 2. Frail patients living in colder temperatures have more rapid progression in frailty. | Hoogendijk et al, 2019 [122]; Chen et al, 2014 [123]; Alakare et al, 2022 [124]; Cesari et al, 2017 [125]; Takauji et al, 2021 [126]; Zhou et al, 2023 [127]; He et al, 2025 [128] |
| Area of focus | Practical interventions and management strategies | Clinical rationale | Supporting references |
|---|---|---|---|
| Screening and measurement | 1. Measure temperature in all clinical assessments of geriatric patients. 2. Use tympanic thermometers for practicality. | Tympanic readings provide a reasonable estimate of core temperature in the elderly and have close proximity to the thermoregulatory center of the brain. | Chen et al, 2020 [43]; Sund-Levander & Grodzinsky, 2020 [31] |
| Nutritional and musculoskeletal health | 1. Ensure adequate caloric and protein intake. 2. Encourage resistance training to maintain muscle mass. 3. Refer to dietitian or physiotherapist for individualized plans. | Malnutrition and sarcopenia lower basal metabolic rate, reduce subcutaneous insulation, and predispose patients to hypothermia and strength-related functional decline. | Grassi et al, 2003 [21]; Kenney & Munce, 2003 [16]; Rogeri et al, 2021 [129]; Covinsky et al, 2003 [130] |
| Co-morbidity management during weather extremes | 1. Have a high suspicion for respiratory infections, hypoglycemia, and blood pressure fluctuations during the winter months for patients with co-morbid diabetes, hypertension, and COPD. 2. Encourage seasonal adaptations, including appropriate clothing, effective home heating and avoiding prolonged exposure to the outdoors during temperature extremes. | Temperature extremes exacerbate chronic disease burden and increase mortality risk in elderly patients with hypertension, diabetes, COPD, and various malignancies. | Qi et al, 2024 [74]; Song et al, 2021 [76]; Yang et al, 2024 [82]; Liu et al, 2023 [131] |
| Medication review and polypharmacy | 1. Review medication lists for common offending agents including antipsychotics, antipyretics, β-blockers, anxiolytics, antidepressants, and opioids. 2. Use antihyperglycemics with low risk of hypoglycemia. 3. Review for drug interactions. | Antipsychotics, antipyretics, β-blockers, anxiolytics, antidepressants, and opioids are well documented to affect thermoregulatory dysfunction. Antihyperglycemics can cause hypoglycemia which often manifests as hypothermia. | Sessler, 2009 [106]; Okada et al, 2024 [107]; Fitzgerald, 1980 [108]; Sund-Levander & Grodzinsky, 2009 [53]; Van Marum et al., 2007 [29] |
| Cognitive and autonomic screening | 1. Evaluate patients with persistent low body temperature for autonomic dysfunction or early cognitive decline. 2. Use validated screening tools to assess for cognitive impairment. 3. Refer to geriatrics if there is concern for neurocognitive disorder. | Low body temperature may be a prodromal marker of a major neurocognitive disorder. | Alagiakrishnan et al., 2023 [119] |