| Lincoff et al [26] | 2024 | Double blind, placebo-controlled trial | 13,970 | Bempedoic acid vs. placebo | 40.6 months | Bempedoic acid reduced major adverse cardiovascular events by 15% compared to placebo. |
| Nicholls et al [27] | 2024 | Randomized, double-blind, placebo-controlled, clinical trial | 13,970 | Bempedoic acid vs. placebo | 3.4 years | Bempedoic acid was associated with a 20% reduction in the total number of major cardiovascular events compared to placebo in high-risk, statin-intolerant patients. |
| Bays et al [28] | 2025 | Subset analysis of randomized, double-blind, placebo-controlled, clinical trial | 13,970 | Bempedoic acid vs. placebo | 40.7 months | Bempedoic acid resulted in a placebo-corrected reduction in LDL-C of 22.5% and hs-CRP of 23.2%, and a 23% reduction in the composite MACE-4 endpoint compared with placebo in people with obesity. |
| Duell et al [29] | 2024 | Pooled patient-level data analysis of two phase 3 randomized clinical trials | 217 | Bempedoic acid vs. placebo | 52 weeks | In patients with HeFH, adding bempedoic acid to maximally tolerated statin therapy resulted in a significant LDL-C reduction of −21.2% at week 12 versus placebo. |
| Taub et al [30] | 2025 | Randomized, double-blind, phase 3 trial | 350 | Inclisiran vs. ezetimibe vs. placebo | 6 months | Inclisiran monotherapy reduced LDL-C by 46.5% at day 150 vs. placebo (1.4% increase) and vs. ezetimibe (−11.2%). |
| Basit et al [31] | 2025 | Meta-analysis of randomized controlled trials | 5,016 | Inclisiran vs. placebo | Trials included up to July 2024 | Inclisiran significantly reduced LDL-C by a mean difference of −50.42% compared with control, without increasing serious adverse event risk. |
| Saad Cleto et al [32] | 2025 | Systematic review and meta-analysis | 30,718 | Inclisiran vs. alirocumab | 24 weeks | Alirocumab demonstrated greater efficacy than inclisiran in enhancing lipid parameters. |
| Rai et al [33] | 2024 | Systematic review and meta-analysis | | Inclisiran | | The meta-analysis found significantly greater reductions in lipid-parameters in HeFH compared to HoFH. |
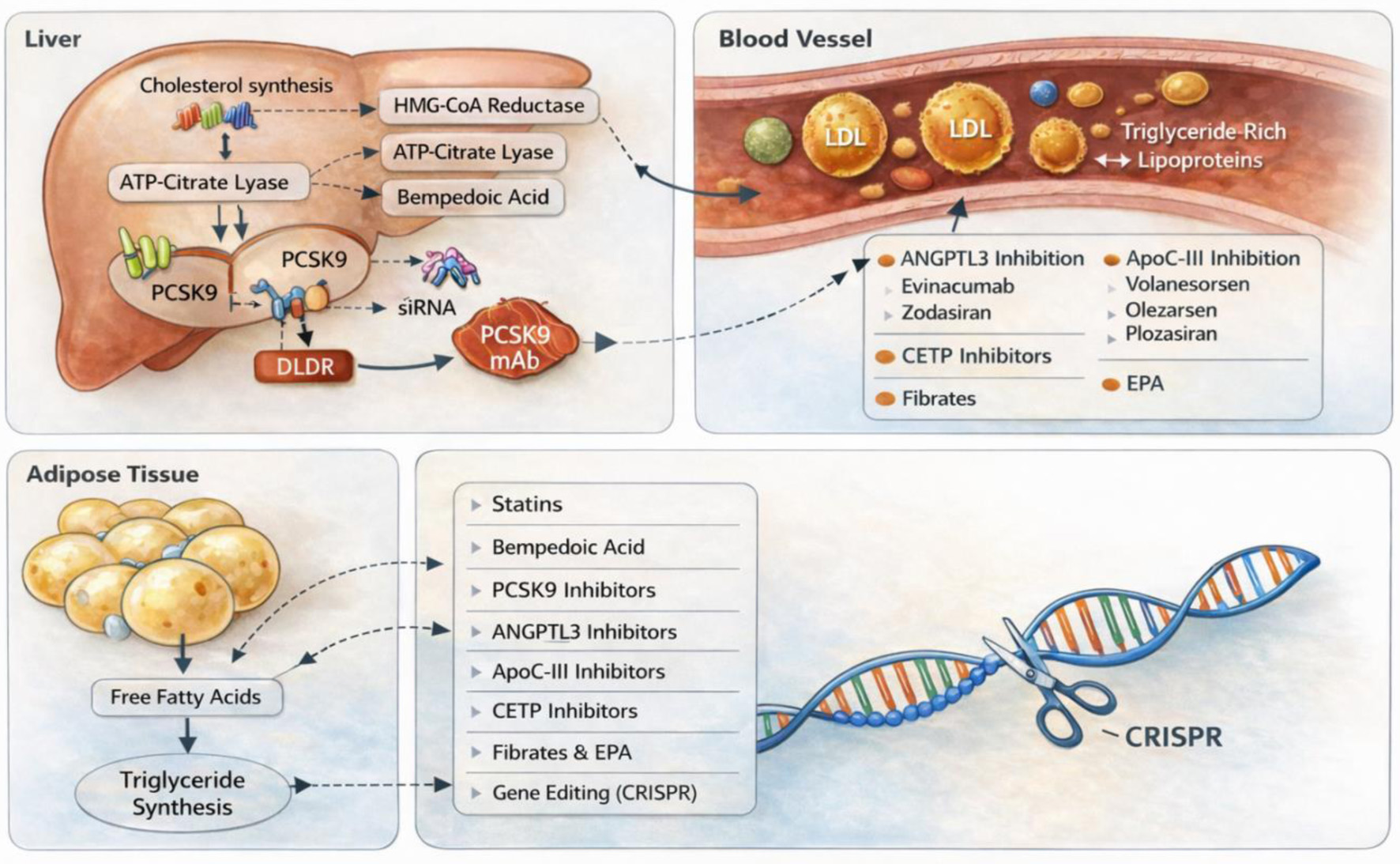
| Rangwala et al [34] | 2024 | Systematic review and meta-analysis | 270 | Evinacumab vs. placebo | | High dose evinacumab significantly reduce lipid parameters with no excess adverse events. |
| Rosenson et al [35] | 2020 | Randomized, double-blind, placebo-controlled Phase 2 clinical trial | 272 | Evinacumab vs. placebo | 16 weeks | Evinacumab reduced LDL-cholesterol by more than 50% at the highest dose compared to placebo. |
| Rosenson et al [36] | 2024 | Phase 2b, randomized, double-blind, placebo-controlled trial | 204 | Zodasiran vs. placebo | 36 weeks | Zodasiran resulted in dose-dependent triglyceride reductions. |
| Kim et al [37] | 2024 | Systematic review and meta-analysis | 53,231 | Fibrate vs. placebo | | Fibrate therapy was linked to a lower incidence of major cardiovascular events, with its beneficial impact primarily attributed to reductions in LDL-cholesterol rather than triglyceride levels. |
| Jakob et al [38] | 2016 | Systematic review and meta-analysis | 16,135 | Fibrate vs. placebo | 4.8 years | Fibrate therapy lowered the combined risk of cardiovascular-disease death, non-fatal myocardial infarction, or non-fatal stroke in primary prevention. |
| Sahebkar et al [40] | 2017 | Systematic review and meta-analysis | 1,388 | Fibrate vs. statin | | Fibrate therapy led to a markedly greater decrease in plasma lipoprotein(a) levels than statin treatment. |
| Gouni-Berthold et al [41] | 2021 | Randomized, double-blind, placebo-controlled phase 3 clinical trial | 114 | Volanesorsen vs. placebo | 26 weeks | Volanesorsen resulted in a 71.2% decrease in triglyceride levels, compared to only a 0.9% change observed in the placebo group. |
| Bergmark et al [42] | 2024 | Randomized, double-blind, placebo-controlled, phase 2b trial | 154 | Olezarsen vs. placebo | 6 months with follow-up up to 12 months | Olezarsen at doses of 50 and 80 mg produced triglyceride reductions of approximately 49.3% and 53.1%, respectively, when compared with the placebo group. |
| Gaudet et al [43] | 2024 | Randomized clinical trial | 226 | Plozasiran vs. placebo | 48 weeks | Plozasiran produced robust triglyceride lowering, with a least-squares mean reduction of 57% at the highest dose; ApoC-III decreased by 77%, and more than 90% of treated participants achieved triglyceride levels below 500 mg/dL. |
| Zhou et al [45] | 2018 | Systematic review and meta-analysis of randomized controlled trials | 34,781 | Anacetrapib vs. placebo | | Anacetrapib significantly improved lipid parameters, increasing HDL-C and reducing LDL-C, non-HDL-C, triglycerides, ApoB, and Lp(a), without a significant increase in adverse events compared with placebo. |
| Kastelein et al [46] | 2024 | Narrative review of phase 2 and phase 3 randomized trials | _ | Obicetrapib vs. placebo/statin | _ | Obicetrapib produced substantial LDL-C reductions of approximately 45–51% in phase 2 studies and 36.3% at day 84 in the phase 3 BROOKLYN trial, with sustained benefit and favorable tolerability. |
| Sayah et al [47] | 2024 | Randomized clinical trial | _ | Icosapent ethyl vs. placebo | 5 years | Icosapent ethyl reduced the primary composite cardiovascular endpoint by 25% and cardiovascular death by 20% in statin-treated patients with elevated triglycerides and high cardiovascular risk. |
| Hafiane et al [48] | 2025 | Phase 3 trial | 17 | Lomitapide vs. baseline | | Lomitapide significantly reduced LDL-C and ApoB in patients with HoFH, with larger reductions at higher doses and associated changes in HDL function. |
| Karwatowska-Prokopczuk et al [49] | 2023 | Randomized, double-blind, placebo-controlled phase 1 study | 29 | Pelacarsen vs. placebo | 204 days | Pelacarsen produced marked dose-dependent reductions in lipoprotein(a), reaching up to approximately 84% with repeated dosing, with acceptable short-term safety. |