| Journal of Clinical Medicine Research, ISSN 1918-3003 print, 1918-3011 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Med Res and Elmer Press Inc |
| Journal website https://jocmr.elmerjournals.com |
Short Communication
Volume 16, Number 10, October 2024, pages 503-508
Elevated Serum Growth Differentiation Factor 15 Levels as a Potential Biomarker of the Efficacy of Imeglimin in Individuals With Type 2 Diabetes Mellitus: An Exploratory Study
Naoki Wadaa, e, Takaaki Murakamia, e, f , Muhammad
Fauzia, Kentaro Sakakia, Shinobu Oshimab, Yoshihito
Shimadab, Kanae Asaib, Ayako Oshimab, Satoko
Nomurab, Erina Joob, Michiko Morib, Ryoko Fujiwarab,
Kenichiro Shideb, Keiko Wadab, Daisuke Yabea
, Nobuya
Inagakic
, Norio Haradaa, d
aDepartment of Diabetes, Endocrinology, and Nutrition, Graduate School of Medicine,
Kyoto University, Kyoto, Japan
bDepartment of Metabolism and Clinical Nutrition,
Kyoto University Hospital, Kyoto, Japan
cMedical Research Institute Kitano
Hospital, PIIF Tazuke-Kofukai, Osaka, Japan
dDepartment of Endocrinology and
Metabolism, School of Medical Sciences, University of Fukui, Japan
eThese
authors contributed equally to this work.
fCorresponding Author: Takaaki
Murakami, Department of Diabetes, Endocrinology and Nutrition, Graduate School of Medicine,
Kyoto University, Shogoin, Sakyo-ku, Kyoto 606-8507, Japan
Manuscript submitted August 19, 2024, accepted October 21, 2024, published online October 30,
2024
Short title: Elevated GDF15 in Treatment With Imeglimin
doi:
https://doi.org/10.14740/jocmr6031
| Abstract | ▴Top |
Background: The aim of the present study was to conduct a prospective observational study to explore the effects of imeglimin on systemic energy metabolism/body composition and to identify potential mitochondria-related biomarkers of the efficacy of the drug in clinical settings.
Methods: In this prospective observational study, 16 participants with type 2 diabetes mellitus in the diabetes clinic of Kyoto University Hospital were enrolled. Individuals were started on imeglimin as monotherapy or add-on therapy.
Results: After 3 months under imeglimin treatment, there was no significant change in basal metabolism or body composition. However, serum levels of growth differentiation factor 15 (GDF15) were higher while those of serum fibroblast growth factor 21 and urine 8-hydroxy-2′-deoxyguanosine were not changed. Additional in vitro examination revealed that imeglimin induces GDF15 protein release from human hepatocytes.
Conclusions: Three-month imeglimin treatment increased serum GDF15 levels in clinical type 2 diabetes mellitus patients along with little change in basal metabolism or body composition, suggesting GDF15 as a potential marker for the efficacy of imeglimin.
Keywords: GDF15; Imeglimin; Diabetes mellitus
| Introduction | ▴Top |
Imeglimin, a recently developed anti-diabetic drug with a structure similar to that of metformin, is reported to improve mitochondrial function and glycemic control in type 2 diabetes mellitus [1]. Imeglimin and metformin both inhibit hepatic gluconeogenesis and stimulate glucose uptake in peripheral tissues, while imeglimin also enhances glucose-induced insulin secretion [2]. Recent studies indicate that imeglimin may preserve beta-cell mass by maintaining or restoring the functional and structural integrity of their mitochondria [3]. However, mitochondria-related biomarkers of systemic energy metabolism have not been investigated in clinical settings.
Growth differentiation factor 15 (GDF15) and fibroblast growth factor 21 (FGF21) are both recognized as biomarkers for diagnosis and severity assessment of mitochondrial diseases including mitochondrial diabetes mellitus [4]. GDF15, a member of the transforming growth factor beta superfamily, is increased by metformin and may mediate its beneficial effect on body weight and energy balance [5]. FGF21, a member of the FGF19 subfamily, reflects insulin sensitivity and energy expenditure [6]. Interestingly, while increased urine 8-hydroxy-2′-deoxyguanosine (8-OHdG) levels have been reported to indicate mitochondrial oxidative damage in clinical and experimental studies [7], imeglimin has been shown to reduce 8-OHimdG levels in diabetes model mice [7].
In this study, we evaluated the mitochondria-related protein levels of GDF15, FGF21, and 8-OhdG, as well as basal metabolism and body composition, in individuals with type 2 diabetes mellitus under imeglimin treatment.
| Materials and Methods | ▴Top |
Study protocol
This prospective observational study was carried out in the diabetes clinic of Kyoto University Hospital (Kyoto, Japan). Individuals with type 2 diabetes mellitus who were started on imeglimin as monotherapy or add-on therapy between March 1, 2022, and September 30, 2022, were enrolled. Inclusion and exclusion criteria are shown here (Supplementary Material 1, jocmr.elmerjournals.com/). Clinical information and data including sex, age, duration of diabetes, diabetic retinopathy and nephropathy, laboratory data, and therapeutic agents before, 3, and 6 months after the initiation of imeglimin (2,000 mg/day) treatment, were collected (Supplementary Material 2, jocmr.elmerjournals.com/). Fasting serum GDF15 (DGD150; R&D System, USA) and FGF21 (DF2100; R&D System, USA), urinary 8-OHdG (Nikken seil Co. Ltd., Japan), basal metabolism by indirect calorimeter (MedGem, Microlife Medical Home Solutions, Inc, USA), and body composition by bioelectrical impedance analysis (InBody 720; InBody Japan Inc., Japan) were evaluated before and 3 months after initiation. The protocol of the study was approved by the Kyoto University Graduate School and Faculty of Medicine, Ethics Committee (R3300, trial registration: UMIN000046316) and conforms to the provisions of the Declaration of Helsinki.
In vitro GDF15 secretion assay
Imeglimin-induced GDF15 secretion from the human hepatocyte cell line HepG2 was evaluated (Supplementary Material 3, jocmr.elmerjournals.com/).
Statistical analysis
Data are presented as average ± standard deviation. Differences between two groups were analyzed by the unpaired Student’s t-test, Wilcoxon test or Fisher’s exact probability test, as appropriate. P value of < 0.05 was considered statistically significant. JMP Pro®, version 17.0 (SAS Institute Inc., NC, USA) was used to carry out the statistical analysis. A scatter plot was made using JMP Pro® version 17.0.
| Results | ▴Top |
Characteristics of the subjects
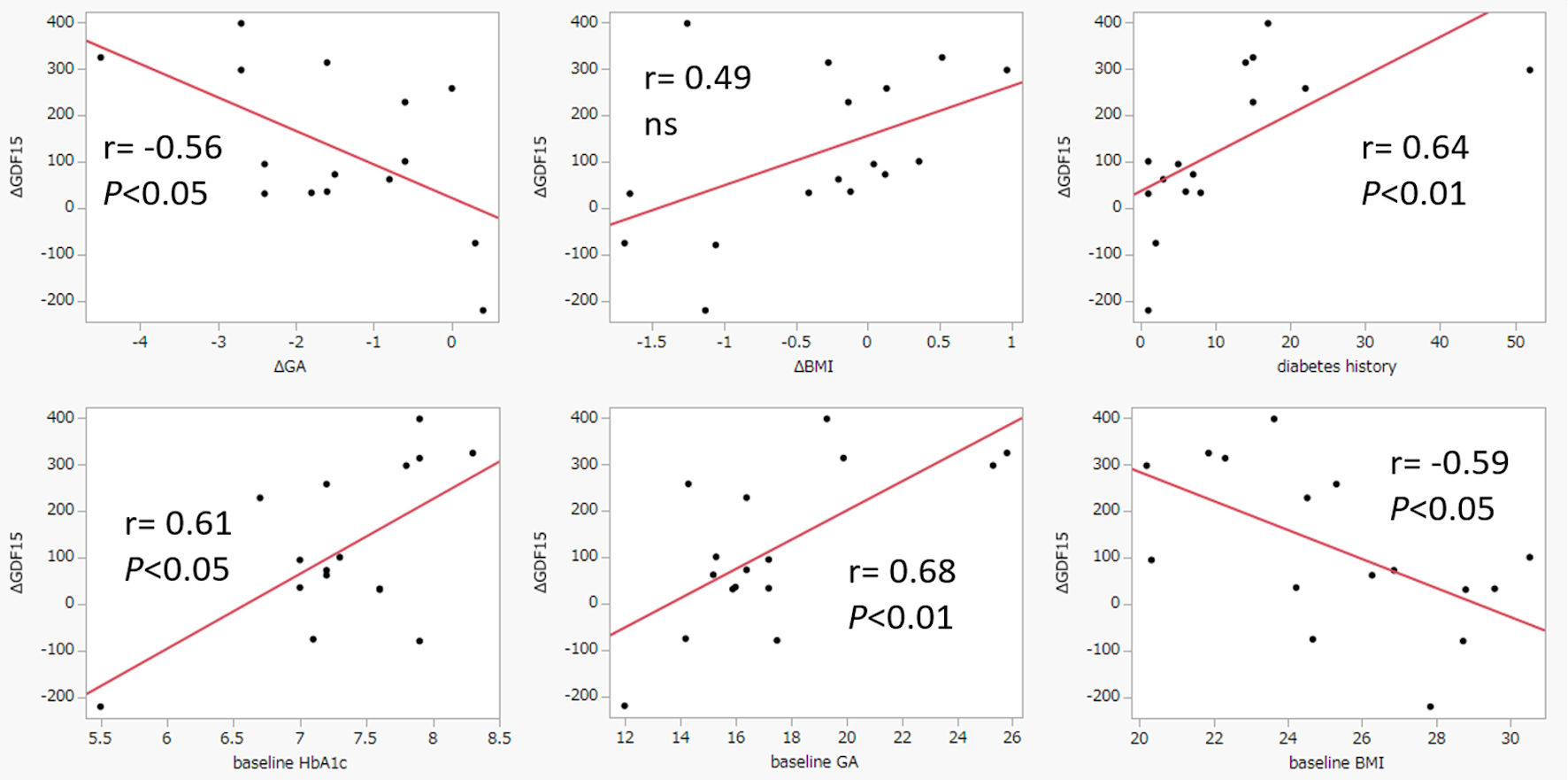
Sixteen individuals with type 2 diabetes mellitus (monotherapy/add-on therapy. n = 2/14) were enrolled (Supplementary Material 2, jocmr.elmerjournals.com/); their profiles are shown in Table 1. After 3 months under imeglimin treatment, glycated hemoglobin (HbA1c) and glycoalbumin (GA) decreased by 0.3% and 1.5%, respectively; the fasting C-peptide index (CPI) and homeostatic model assessment of beta cell function (HOMA-β) increased by 0.7 and 22.9, respectively. No significant changes in homeostatic model assessment for insulin resistance (HOMA-IR), quantitative insulin sensitivity check index (QUICKI), lipids, renal function, or liver function were observed. Body fat percentage and resting metabolic rate did not significantly change. GDF15 significantly increased (P < 0.05), whereas no significant changes in FGF21 or urine 8-OHdG were observed. The changes in GDF15 during 3-month treatment with imeglimin (ΔGDF15) demonstrated a significant negative correlation with those in GA levels (r = -0.56, P < 0.05) (Fig. 1), whereas ΔGDF15 had no significant correlation with Δbody mass index (BMI). ΔGDF15 had significant positive correlations with diabetes history, baseline HbA1c and GA levels, respectively, whereas ΔGDF15 showed a significant negative correlation with baseline BMI (Fig. 1). ΔGDF15 tended to be negatively correlated with ΔHbA1c (r = -0.23), although the difference was not statistically significant (P = 0.40). Interestingly, the nine patients who were taking metformin at baseline tended to have higher ΔGDF15 than the seven patients who were not taking metformin (149.9 ± 155.5 ng/mL vs. 75.2 ± 192.2 ng/mL, P = 0.42), although the differences did not reach statistical significance. Clinical follow-up data of 13 of the 16 patients were obtained after 6 months: HbA1c was 0.5% lower and glycoalbumin was 1.5% lower in these patients. Adverse effects during the observation period are shown in Table 1.
 Click to view |
Table 1. Profile of the Subjects and Laboratory
Data Before, 3, and 6 Months After Initiation of Imeglimin Treatment, and
Adverse Effects of Imeglimin During the Study |
 Click for large image |
Figure 1. Correlation between Δgrowth differentiation factor 15 (GDF15) and Δglycoalbumin (GA), Δbody mass index (BMI), diabetes history, baseline HbA1c, baseline GA, baseline BMI. Correlation between ΔGDF15 and each parameter is shown as “r”. ΔGDF15 vs. each parameter, *P < 0.05. ns: not significant; HbA1c: glycated hemoglobin. |
Effects of imeglimin in HepG2 cells
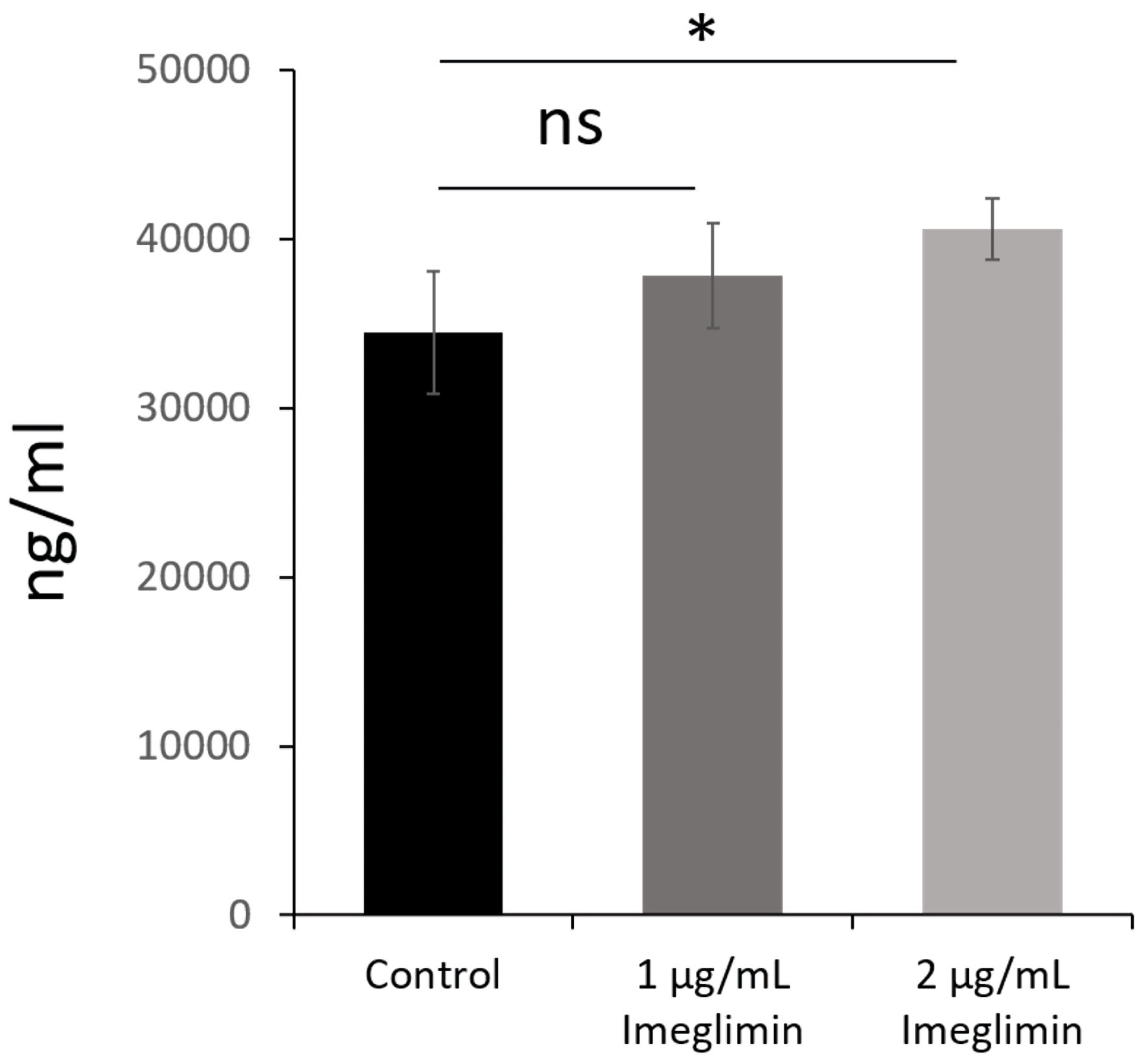
Imeglimin-induced GDF15 release from HepG2 cells was evaluated. After 48 h incubation, imeglimin-treated HepG2 cells released a higher concentration of GDF15 compared to that of control, in a dose-dependent manner (Fig. 2).
 Click for large image |
Figure 2. GDF15 released from imeglimin-treated HepG2 cells. Control (n = 3), 1 µg/mL imeglimin-treated (n = 3), 2 µg/mL imeglimin-treated HepG2 cells (n = 3). Data are mean ± SD. Control vs. imeglimin-treated, *P < 0.05, ns: not significant; SD: standard deviation. |
| Discussion | ▴Top |
Imeglimin as monotherapy or add-on therapy resulted in improved glycemic control and β-cell function-related indices (fasting CPI and HOMA-β) in a clinical setting. Increased serum GDF15 levels were noted, but there were no significant changes in serum FGF21, urine 8-OHdG, or basal metabolism and body composition.
GDF15 has been reported to be a key molecule in the beneficial effects of metformin on glycemic control and energy metabolism via hepatocytes and intestinal cells [5]. A genome-wide association study suggested that elevated GDF15 expression across various tissues may contribute to improved glycemic control in type 2 diabetes patients taking metformin [8]. Consistent with their similar chemical structure, a recent study found GDF15 mRNA expression in cultured hepatocytes to be notably increased by imeglimin [9]. Moreover, the additional GDF15 protein would be supplied at least partially from hepatocytes via the in vitro experiment conducted at the concentration of imeglimin which was close to the serum maximum concentration after the 7-day clinical administration according to the interview form [10].
Interestingly, ΔGDF15 showed significant negative correlations with ΔGA in the present study. This finding further suggests GDF15 as a potential biomarker for the efficacy of imeglimin in individuals with type 2 diabetes mellitus. Indeed, it has been reported that GA assesses short-term efficacy of imeglimin more accurately than HbA1c [11]. In the present study, diabetes history, baseline HbA1c and GA levels, and baseline BMI showed significant correlations with ΔGDF15, which may have implications regarding the mechanism underlying the differing responses to imeglimin of patients with type 2 diabetes mellitus [12-15]. In fact, participants with longer diabetes history showed significantly higher GA levels at baseline. As ΔGDF15 also had a significant positive correlation with baseline GA, the GA levels of the subjects with longer diabetes history might underlie the relationship between ΔGDF15 and diabetes history noted in this study. Further investigation is required to establish assessment of the efficacy of imeglimin based on GDF15 values.
Additionally, the present study noted no significant change in basal metabolism during imeglimin treatment, which is consistent with a previous report [12]. We also found no significant influence of imeglimin on body composition.
This exploratory study has several limitations including the relatively small number of subjects, which limits the generalizability of the findings. In addition, our 3-month treatment period is insufficient to assess the long-term effects of the drug, particularly in terms of the sustainability of the GDF15 levels and the metabolic changes. In fact, in a previous report [5], the association between change in body weight and change in plasma GDF15 between 0 and 18 months among metformin-treated participants were assessed. In the future, longer-term, direct comparisons between imeglimin and other established treatments for type 2 diabetes are warranted. Furthermore, inclusion of patients with various degrees of renal or hepatic impairment would provide insights into the broader applicability of imeglimin.
In conclusion, 3-month imeglimin treatment increased serum GDF15 levels in clinical type 2 diabetes mellitus patients along with little change in basal metabolism or body composition, suggesting GDF15 as a potential marker for the efficacy of imeglimin.
| Supplementary Material | ▴Top |
Suppl 1. Selection and exclusion criteria.
Suppl 2. Time course of the study. Three participants were transferred to a family clinic between 3 and 6 months.
Suppl 3. Cell culture and in vitro GDF15 secretion assay.
Acknowledgments
None to declare.
Financial Disclosure
NI received joint research grants from Daiichi Sankyo, Terumo, and Drawbridge Health, received speaker honoraria from Kowa, MSD, Astellas Pharma, Novo Nordisk Pharma, Ono, Nippon Boehringer Ingelheim, Takeda, and Mitsubishi Tanabe Pharma, and received scholarship grants from Kissei, Sanofi, Daiichi-Sankyo, Mitsubishi Tanabe, Takeda, Japan Tobacco, Kyowa Kirin, Sumitomo Pharma, Astellas Pharma, MSD, Eli Lilly Japan, Ono, Sanwa Kagaku Kenkyusho, Nippon Boehringer Ingelheim, Novo Nordisk Pharma, Novartis Pharma, Teijin Pharma, and Life Scan Japan. DY has received consulting or speaker fees from Sumitomo Pharma Co., Ltd., Eli Lilly Japan K.K., MSD K.K., Novo Nordisk Pharma Ltd., Nippon Boehringer Ingelheim Co., Ltd., and Tanabe Mitsubishi Pharma, Co., Ltd., and clinically commissioned/joint research grants from Taisho Pharmaceutical Co., Ltd., Novo Nordisk Pharma Ltd., Arklay Co. Ltd. and Nippon Boehringer Ingelheim Co., Ltd. NH received scholarship grants from Mitsubishi Tanabe Pharma Co., Ltd., Ono Pharmaceutical Co., Ltd., Nippon Boehringer Ingelheim Co., Ltd., and Sanofi K.K. TM received a joint research grant from Sumitomo Pharma. The remaining authors have no conflicts of interest to disclose.
Conflict of Interest
None to declare.
Informed Consent
The written informed consent was obtained from the subject.
Author Contributions
NW contributed to data collection, analysis, discussions of this study and wrote the manuscript. TM contributed to planning, design, data collection, analysis, discussions, and editing the manuscript. MF contributed to performing in vitro experiments. KS (Kentaro Sakaki), SO, YS, KA, AO, SN, EJ, MM, RF contributed to collecting clinical data. KS (Kenichiro Shide) and KW contributed to planning and discussions. DY and NI contributed to discussions and editing the manuscript. NH contributed to planning, design, discussions and editing the manuscript. All authors contributed to the article and approved the submitted version.
Data Availability
The data supporting the findings of this study are available from the corresponding author on reasonable request.
Abbreviations
GDF15: growth differentiation factor 15; FGF21: fibroblast growth factor 21; 8-OhdG: urine 8-hydroxy-2′-deoxyguanosine; GA: glycoalbumin; CPI: C-peptide index
| References | ▴Top |
- Fauzi M, Murakami T, Yabe D, Inagaki N. Current
understanding of imeglimin action on pancreatic beta-cells: Involvement of mitochondria and
endoplasmic reticulum homeostasis. J Diabetes Investig. 2023;14(2):186-188.
doi pubmed - Yingyue Q, Sugawara K, Takahashi H, Yokoi N, Ohbayashi K,
Iwasaki Y, Seino S, et al. Stimulatory effect of imeglimin on incretin secretion.
J Diabetes Investig. 2023;14(6):746-755.
doi pubmed - Fauzi M, Murakami T, Fujimoto H, Botagarova A, Sakaki K,
Kiyobayashi S, Ogura M, et al. Preservation effect of imeglimin on pancreatic beta-cell mass:
Noninvasive evaluation using (111)In-exendin-4 SPECT/CT imaging and the perspective of
mitochondrial involvements. Front Endocrinol (Lausanne). 2022;13:1010825.
doi pubmed - Murakami T, Ueba Y, Shinoto Y, Koga Y, Kaneda D, Hatoko T,
Kato T, et al. Successful Glycemic Control Decreases the Elevated Serum FGF21 Level without
Affecting Normal Serum GDF15 Levels in a Patient with Mitochondrial Diabetes.
Tohoku J Exp Med. 2016;239(2):89-94.
doi pubmed - Coll AP, Chen M, Taskar P, Rimmington D, Patel S, Tadross
JA, Cimino I, et al. GDF15 mediates the effects of metformin on body weight and energy balance.
Nature. 2020;578(7795):444-448.
doi pubmed - Tomlinson E, Fu L, John L, Hultgren B, Huang X, Renz M,
Stephan JP, et al. Transgenic mice expressing human fibroblast growth factor-19 display
increased metabolic rate and decreased adiposity. Endocrinology. 2002;143(5):1741-1747.
doi pubmed - Sanada J, Obata A, Fushimi Y, Kimura T, Shimoda M, Ikeda T,
Nogami Y, et al. Imeglimin exerts favorable effects on pancreatic beta-cells by improving
morphology in mitochondria and increasing the number of insulin granules. Sci Rep.
2022;12(1):13220.
doi pubmed - Sakaue S, Kanai M, Tanigawa Y, Karjalainen J, Kurki M,
Koshiba S, Narita A, et al. A cross-population atlas of genetic associations for 220 human
phenotypes. Nat Genet. 2021;53(10):1415-1424.
doi pubmed - Hozumi K, Sugawara K, Ishihara T, Ishihara N, Ogawa W.
Effects of imeglimin on mitochondrial function, AMPK activity, and gene expression in
hepatocytes. Sci Rep. 2023;13(1):746.
doi pubmed - Sumitomo Pharma Co. Twymeeg® Tablets Ethical Drugs Attachment, 3rd Edition. Revised April 2022. p. 34-36. https://www.pmda.go.jp/files/000246722.pdf. August 17, 2024 available.
- Osonoi T, Shirabe S, Saito M, Hosoya M, Douguchi S, Ofuchi
K, Katoh M. Comparative evaluation of clinical glycemic control markers treated with imeglimin
and its effect on erythrocytes in patients with type 2 diabetes mellitus: study protocol of a
single-arm, open-label, prospective, exploratory trial. Front Pharmacol.
2023;14:1205021.
doi pubmed - Dubourg J, Fouqueray P, Thang C, Grouin JM, Ueki K. Efficacy
and Safety of Imeglimin Monotherapy Versus Placebo in Japanese Patients With Type 2 Diabetes
(TIMES 1): A Double-Blind, Randomized, Placebo-Controlled, Parallel-Group, Multicenter Phase 3
Trial. Diabetes Care. 2021;44(4):952-959.
doi pubmed - Hagi K, Kochi K, Watada H, Kaku K, Ueki K. Factors
contributing to the clinical effectiveness of imeglimin monotherapy in Japanese patients with
type 2 diabetes mellitus. J Diabetes Investig. 2024;15(9):1239-1247.
doi pubmed - Hagi K, Kochi K, Watada H, Kaku K, Ueki K. Differences in
imeglimin response in subgroups of patients with type 2 diabetes stratified by data-driven
cluster analysis: A post-hoc analysis of imeglimin clinical trial data. Diabetes Obes Metab.
2024;26(9):3732-3742.
doi pubmed - Hagi K, Kochi K, Watada H, Kaku K, Ueki K. Effect of patient
characteristics on the efficacy and safety of imeglimin monotherapy in Japanese patients with
type 2 diabetes mellitus: A post-hoc analysis of two randomized, placebo-controlled trials.
J Diabetes Investig. 2023;14(9):1101-1109.
doi pubmed
This
article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0
International License, which permits unrestricted non-commercial use, distribution, and
reproduction in any medium, provided the original work is properly cited.
Journal
of Clinical Medicine Research is published by Elmer Press Inc.