| Journal of Clinical Medicine Research, ISSN 1918-3003 print, 1918-3011 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Med Res and Elmer Press Inc |
| Journal website https://jocmr.elmerjournals.com |
Case Report
Volume 16, Number 10, October 2024, pages 509-517
Addition of Sacubitril/Valsartan to Mineralocorticoid Receptor Antagonist Therapy in Primary Aldosteronism: Effects on Plasma Aldosterone Concentration and Plasma Renin Activity
Keisuke Okamuraa, b, d, Masatoshi Matsushimac, Yosuke Takamiyab, c, Tetsu Okudab, Hideto Sakoa, Akihiro Udoa, Kenichiro Taniguchia, Shogo Morisakia, Ichiro Imamuraa, Hidenori Uratac, Shin-ichiro Miurab
aDepartment of Cardiology and Cardiovascular Center, Imamura Hospital, Tosu, Saga,
Japan
bDepartment of Cardiology, Fukuoka University School of Medicine, Fukuoka,
Fukuoka, Japan
cDepartment of Cardiovascular Diseases, Fukuoka University
Chikushi Hospital, Chikushino, Fukuoka, Japan
dCorresponding Author: Keisuke
Okamura, Department of Cardiology and Cardiovascular Center, Imamura Hospital, 1523-6,
Todoroki-machi, Tosu, Saga 841-0061, Japan
Manuscript submitted September 4, 2024, accepted October 17, 2024, published online October 30,
2024
Short title: Sacubitril/Valsartan Therapy in PA
doi:
https://doi.org/10.14740/jocmr6058
| Abstract | ▴Top |
In the pharmacologic treatment of primary aldosteronism (PA), titration of mineralocorticoid receptor antagonist (MRA) dosing is necessary to reverse the renin suppression caused by high aldosterone levels. However, we often encounter cases in which the plasma renin activity (PRA) does not achieve the target level, even with the maximum dose of MRA. In this setting, sacubitril/valsartan, a combination of a neprilysin inhibitor and an angiotensin II type 1 receptor blocker that is approved for use as adjunctive therapy with an MRA, has been reported to inhibit aldosterone secretion both in vitro and in vivo. If sacubitril/valsartan proves to be effective in this context, it may offer a promising treatment for PA. However, there are few reports on the use of sacubitril/valsartan in this disease. We used add-on sacubitril/valsartan in three patients with PA, in whom blood pressure was insufficiently reduced and PRA remained suppressed despite administering the maximum dose of MRA. With the addition of sacubitril/valsartan, the decrease in plasma aldosterone concentration (PAC) was more marked than the increase in PRA. Because MRAs do not suppress aldosterone production but instead act by blocking mineralocorticoid receptors, use of these agents actually promotes the renin-angiotensin system and leads to increased PAC resulting from positive feedback. The pathological significance of the phenomenon whereby PAC increases with MRA administration but decreases with the addition of sacubitril/valsartan is unclear. In PA, more effective treatment may be possible by suppressing aldosterone with sacubitril/valsartan and blocking the action of aldosterone with MRAs.
Keywords: Primary aldosteronism; Sacubitril/valsartan; Renin; Aldosterone; Mineralocorticoid receptor antagonists; Hypertension
| Introduction | ▴Top |
Primary aldosteronism (PA) is a major form of secondary hypertension that causes cardiovascular events independent of blood pressure (BP) elevation [1, 2]. PA can result in fatal complications, and surgical treatment may be strongly recommended [3]. PA encompasses several subtypes, including aldosterone-producing adenoma (APA), which can be confirmed by computed tomography, aldosterone-producing microadenoma, and idiopathic hyperaldosteronism (IHA). Additionally, there are nonfunctioning adenomas that do not produce aldosterone [4].
For these reasons, it is necessary to confirm the unilaterality of PA by adrenal vein sampling (AVS) before definitive surgical treatment [5, 6]. In the context of treatment for PA, it was originally thought that prognosis did not differ with surgical vs. pharmacologic therapy [7], although it has recently been reported that surgical therapy is associated with a better prognosis, as compared to inadequate pharmacologic therapy [8, 9]. However, if the patient requests pharmacologic therapy or if AVS shows bilateral lesions, treatment with a mineralocorticoid receptor antagonist (MRA) becomes necessary. In pharmacologic therapy for PA, it is important to reverse the suppression of renin caused by high aldosterone levels, and titration of the medication dose is considered necessary to achieve a plasma renin activity (PRA) level greater than 1.0 ng/mL/h [9]. However, we often encounter cases in which the PRA does not achieve this target level, even with the maximum dose of MRA [10]. If MRA monotherapy cannot sufficiently reverse the inhibition of PRA in PA, the level of patient risk has not been sufficiently mitigated; however, concomitant use of additional MRAs is usually contraindicated.
In the setting of unsuccessful MRA monotherapy for PA, sacubitril/valsartan (an angiotensin II type 1 receptor-neprilysin inhibitor) has been reported to suppress aldosterone secretion both in vitro and in vivo [11-14]. Sacubitril/valsartan is a combination drug composed of valsartan, an angiotensin receptor blocker (ARB), and sacubitril, an inhibitor of neprilysin, a natriuretic peptide (NP)-degrading enzyme. If sacubitril/valsartan, which is approved to treat hypertension and for use as adjunctive therapy with an MRA, proves to be effective in this context, this medication may offer a very promising treatment option for PA. However, there are few reports on the use of sacubitril/valsartan in patients with PA. Here, we report on changes in laboratory values after adding sacubitril/valsartan in three patients with PA whose BP was insufficiently reduced and whose PRA remained suppressed despite administering the maximum dose of an MRA.
| Case Reports | ▴Top |
Case 1
This female patient was in her 40s and had juvenile hypertension, hypokalemia, a 19-mm left adrenal mass, and a high aldosterone-to-renin ratio (ARR). She was diagnosed with PA on the basis of positive results on a captopril challenge test, furosemide stress test, and saline infusion test.
Because the patient requested radical surgical treatment, “super-selective” segmental AVS using a microcatheter was performed after adrenocorticotropic hormone loading to confirm detailed mapping of lesions within the adrenal glands [15, 16]. We attempted to advance the microcatheter into both branches of the adrenal vein [17], and we were able to confirm the left adrenal mass using contrast imaging and collected blood at various locations. However, in the right adrenal gland, the vascular lumen collapsed as a result of narrowing of the right adrenal vein due to hypoplasia of the gland. Therefore, although the microcatheter was successfully inserted, it was impossible to collect blood.
The results of AVS suggested that PA in this patient was due to IHA, a left APA, or bilateral APAs [18]. Regardless of an APA on the left side, the right adrenal gland was hypoplastic, indicating that removal of the left adrenal gland may lead to adrenal insufficiency or decline. After consulting with the patient, we decided not to perform AVS again but rather to proceed with pharmacologic therapy.
As BP in this patient was not adequately lowered with the originally prescribed calcium channel blocker (amlodipine 5 mg), an MRA (esaxerenone) was added, and the MRA dose was increased to the maximum (5 mg) (Fig. 1). Even after adding esaxerenone, the BP did not decrease sufficiently. PRA temporarily rose to 1.3 ng/mL/h and then fell below 1.0 ng/mL/h. Aldosterone increased by approximately threefold. Therefore, we switched from amlodipine to sacubitril/valsartan and observed both the PRA and plasma aldosterone concentration (PAC); PRA temporarily rose to 1.5 ng/mL/h but then decreased, and the PAC gradually decreased.
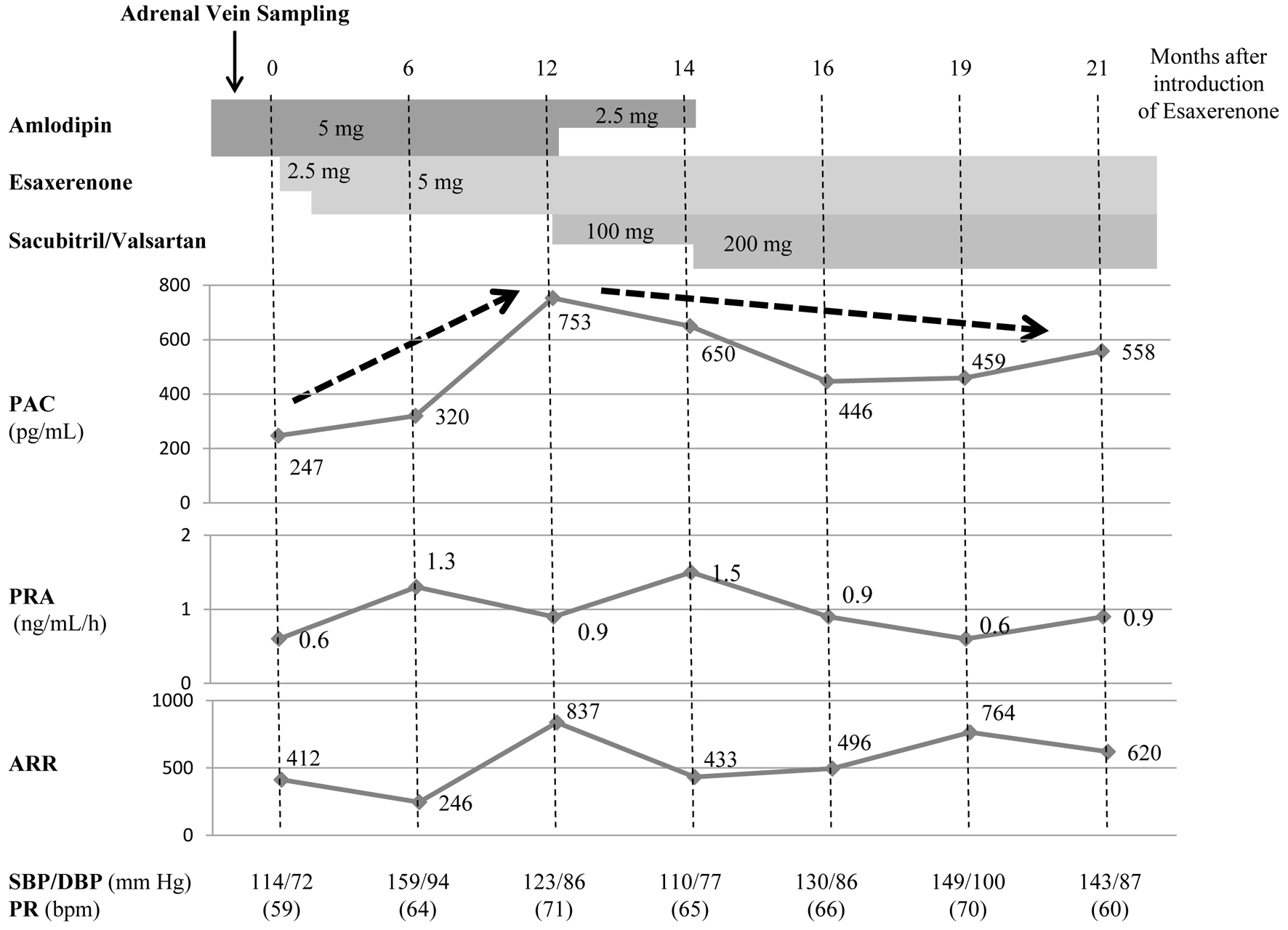 Click for large image |
Figure 1. Case 1 involved a patient with primary aldosteronism likely caused by an aldosterone-producing adenoma (calcium channel blocker → sacubitril/valsartan). The patient was on amlodipine, with insufficient reduction in BP. Therefore, esaxerenone was added, although BP still did not decrease sufficiently, and the PAC increased. Amlodipine was subsequently changed to sacubitril/valsartan; PRA temporarily increased, and the PAC gradually decreased. PAC: plasma aldosterone concentration; PRA: plasma renin activity; ARR: aldosterone-to-renin ratio; SBP: systolic blood pressure; DBP: diastolic blood pressure; PR: pulse rate; bpm: beats per minute. |
Case 2
This female patient was in her 60s and had a history of hypertension diagnosed in her 40s. Her ARR was high. A captopril challenge test was positive, confirming the diagnosis of PA. No mass was found in the adrenal glands, and IHA or a microadenoma was suspected.
As the patient preferred pharmacologic therapy, the maximum dose of esaxerenone (5 mg) was added to the amlodipine 5 mg that she was already taking (Fig. 2). With the addition of esaxerenone, the PAC increased, PRA remained below 1.0 ng/mL/h, and BP did not decrease sufficiently. Over the 3 years following the addition of esaxerenone, the PAC gradually decreased. Thereafter, amlodipine was switched to sacubitril/valsartan, and mild reductions in the PAC and ARR were observed.
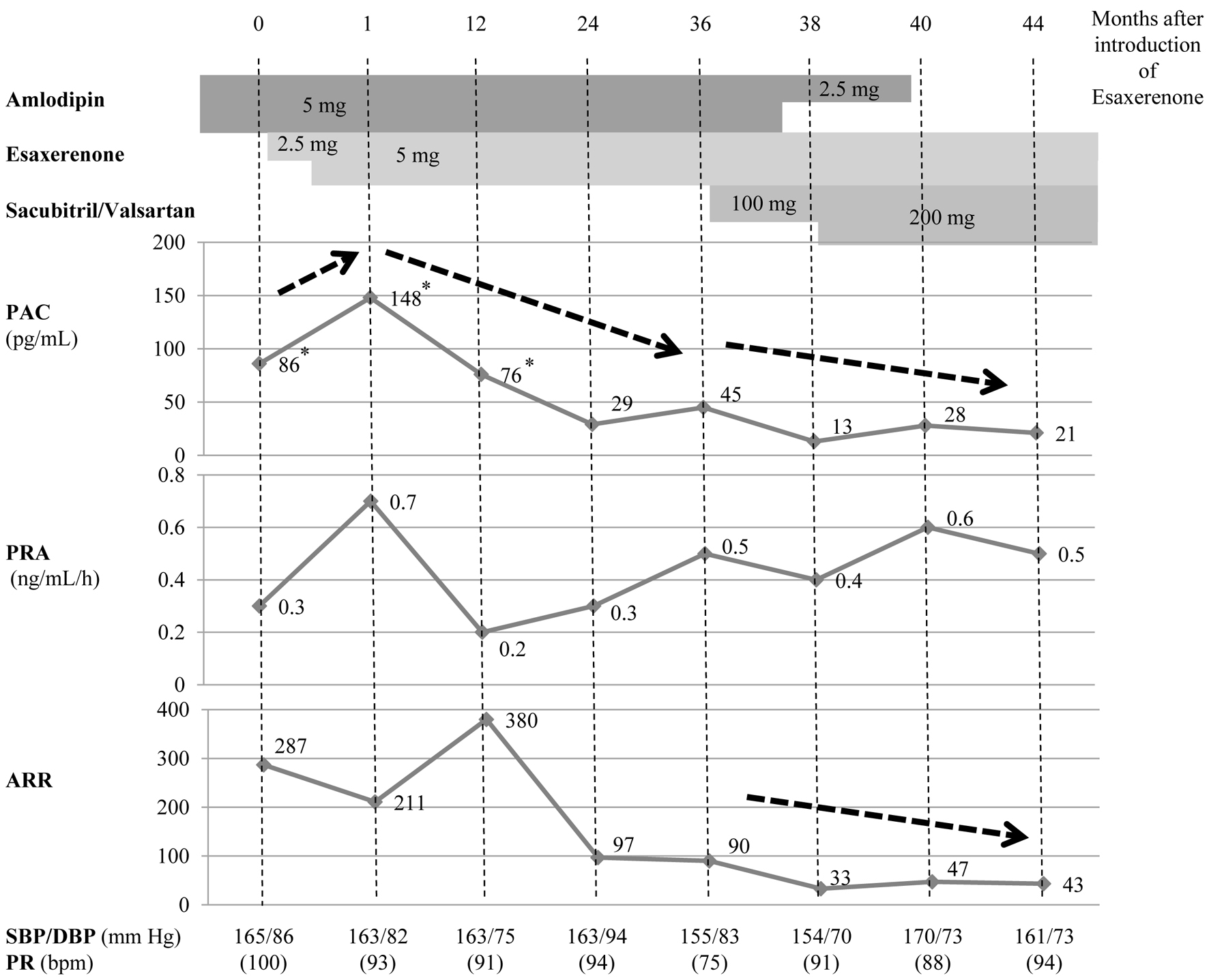 Click for large image |
Figure 2. Case 2 involved a patient with primary aldosteronism likely caused by idiopathic hyperaldosteronism (calcium channel blocker → sacubitril/valsartan). Although the addition of esaxerenone led to an increased PAC, PRA did not sufficiently increase. About 3 years after esaxerenone was added, the PAC gradually decreased. After switching amlodipine to sacubitril/valsartan, the PAC and ARR decreased slightly. *PAC values measured by radioimmunoassay prior to March 2021 have been converted to values consistent with the use of CLEIA to reflect changes in the standard assay used to measure PAC [19]. PAC: plasma aldosterone concentration; PRA: plasma renin activity; ARR: aldosterone-to-renin ratio; CLEIA: chemiluminescent enzyme immunoassay. |
Case 3
This female patient was in her 30s and was diagnosed with PA on the basis of early-onset hypertension, a high ARR, and a positive captopril challenge test. She had no obvious adrenal adenoma. Given that she experienced systemic itching and edema due to the contrast agent used for contrast-enhanced computed tomography, she chose pharmacologic treatment without AVS.
Poor BP control, hypokalemia, and hyporeninemia persisted despite administration of spironolactone 50 mg. Therefore, eplerenone 100 mg was carefully added to MRA therapy (Fig. 3). However, no improvement was observed. Therefore, eplerenone was replaced with esaxerenone 5 mg, which is reported to have strong mineralocorticoid receptor selectivity. Nevertheless, PRA did not exceed 1.0 ng/mL/h.
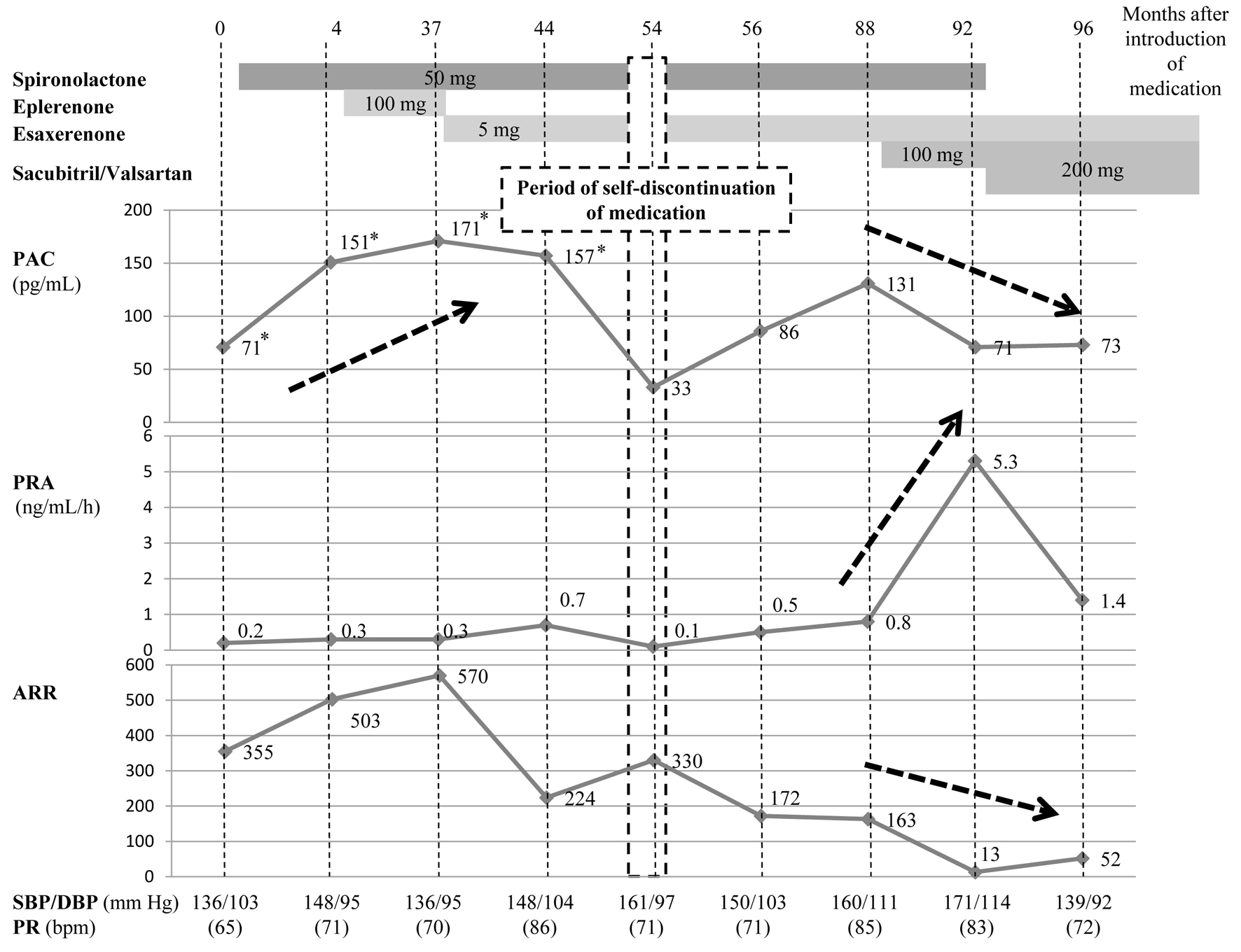 Click for large image |
Figure 3. Case 3 involved a patient with primary aldosteronism likely caused by idiopathic hyperaldosteronism (spironolactone → sacubitril/valsartan). Although eplerenone was changed to esaxerenone, PRA did not exceed 1.0 ng/mL/h. After the patient temporarily self-discontinued treatment, the PAC (which had been elevated with mineralocorticoid receptor antagonist decreased, and PRA was again suppressed as a result of primary aldosteronism. After the medication regimen was resumed, sacubitril/valsartan was added. Because PRA markedly increased and the PAC decreased, spironolactone was discontinued, and sacubitril/valsartan was increased to the maximum dose. Finally, the PAC and ARR decreased, and PRA remained above 1.0 ng/mL/h. PAC: plasma aldosterone concentration; PRA: plasma renin activity; ARR: aldosterone-to-renin ratio. |
The patient temporarily self-discontinued treatment for 2 weeks, and blood tests revealed that the PAC, which had been elevated during MRA therapy, decreased. The medication regimen was resumed, but BP control gradually became poor; thus, sacubitril/valsartan 100 mg was added. Because PRA markedly increased and the PAC decreased, spironolactone was discontinued, and the dose of sacubitril/valsartan was increased to 200 mg. PRA then exceeded 1.0 ng/mL/h.
Postscript
In March 2021, during the course of follow-up in case 2 and case 3, the standard assay used to measure PAC changed from radioimmunoassay to chemiluminescent enzyme immunoassay (CLEIA) [19].
| Discussion | ▴Top |
In this report, we present three cases in which sacubitril/valsartan was added to pharmacologic treatment in patients with PA who had already received therapy with an MRA. Case 1 involved a suspected APA in a patient receiving an MRA. Case 2 involved suspected IHA in a patient receiving an MRA. Case 3 involved suspected IHA in a patient receiving two types of MRAs.
Changes in PRA and PAC
In all cases, we intended to reverse the inhibition of PRA by titrating the MRA dose so that PRA exceeded 1.0 ng/mL/h [9]. Even with the maximum dose of MRA, however, PRA remained less than 1.0 ng/mL/h, and pharmacologic therapy was considered insufficient. With the addition of sacubitril/valsartan, no obvious changes in PRA were seen in case 1 and case 2, although a significant increase in PRA was observed in case 3. By contrast, marked reductions in PAC occurred in all cases.
MRAs, such as spironolactone, generally exert their effect not by suppressing aldosterone production but rather by blocking the mineralocorticoid receptor, causing activation of the renin-angiotensin-aldosterone system (RAAS) and thus resulting in increased PAC due to positive feedback [20]. In patients with PA, administration of MRAs normalizes the suppressed PRA, and PAC increases [20]. The varying degrees of inhibition of mineralocorticoid receptor binding by type of MRA have been demonstrated in vitro [21], and it has been reported that the effects of MRAs may differ across patients [10]. However, the in vivo pathological significance of increased PAC due to administration of MRAs is unknown.
In case 3, PRA significantly increased after administration of sacubitril/valsartan. Aldosterone production, which was enhanced as a result of strong positive feedback due to the two MRA drugs, may have been suppressed by the addition of sacubitril/valsartan, facilitating an increase in PRA. Blocking the RAAS often causes unexpected problems in vivo, such as the aldosterone breakthrough phenomenon [22]. In the treatment of PA, it may be beneficial not only to block mineralocorticoid receptors by using MRAs but also to directly suppress aldosterone production in the adrenal glands.
In case 2, the PAC appeared to increase and then gradually decrease after long-term administration of esaxerenone prior to the addition of sacubitril/valsartan. Long-term administration of spironolactone has been reported to cure IHA [23] and to suppress excessive aldosterone secretion caused by an APA [24]. In this patient, the PAC may have decreased as a result of the long-term effects of the MRA.
Increased intake of salt suppresses renin secretion; thus, it can be difficult to assess the effectiveness of MRA treatment by using PRA as a surrogate marker in patients with PA [25]. PRA is an unstable marker because it is based on measuring enzyme activity, for which measured values can change depending on the blood collection environment and storage conditions.
Even in the cases presented here, it was difficult to perform longitudinal observation based on PRA, as the transition of PRA is unstable. It would be preferable to evaluate pathophysiology on the basis of PAC rather than PRA. Reduction in PAC with the addition of sacubitril/valsartan seems to be a common phenomenon, although the pathological significance of this finding is unclear.
NPs and suppression of aldosterone secretion
NPs, such as atrial NP and B-type NP, have been reported to have vasodilatory effects, natriuretic effects, and aldosterone secretion-suppressing effects via the action of cyclic guanosine monophosphate (cGMP), a plasma biomarker that inhibits the RAAS [26]. NP binds to the NP receptor A that is expressed on the surface of the cell membrane of both smooth muscle cells and endothelial cells in blood vessels throughout the body. NP receptor A suppresses secretion of aldosterone from the adrenal glands by increasing intracellular cGMP and activating the G-kinase system [27].
Neprilysin is an endopeptidase that degrades NPs [28]. Neprilysin is present in many organs, including the proximal renal tubule, heart, brain, lungs, and vascular endothelium. In humans, inhibition of neprilysin promotes natriuresis; suppresses the sympathetic nervous system; reduces plasma vasopressin, PAC, and PRA; and improves the hemodynamic profile in patients with heart failure with reduced ejection fraction (HFrEF) [29]. Sacubitril suppresses the endogenous degradation of NP by inhibiting neprilysin. As NPs inhibit secretion of aldosterone, sacubitril is thought to suppress the organ-damaging effects of aldosterone. However, neprilysin is involved in the degradation of not only NP but also other peptides, such as angiotensin II, adrenomedullin, bradykinin, and endothelin. If a neprilysin inhibitor is used alone, levels of angiotensin II increase, worsening the pathophysiological condition. Therefore, sacubitril/valsartan has been used clinically in combination with an RAAS inhibitor.
Sacubitril/valsartan and aldosterone
In a canine RAAS activation model with a low-salt diet, sacubitril/valsartan increased circulating levels of cGMP and decreased PAC [12]. Administration of sacubitril/valsartan in healthy human participants has also been shown to suppress PAC and increase PRA [30].
Overall, sacubitril/valsartan inhibits PAC in vivo. Administration of sacubitril/valsartan in patients with HFrEF has been reported to result in increased neprilysin, cGMP, and PRA and significantly reduced PAC and plasma N-terminal pro-B-type NP levels [13]. The aldosterone-lowering effect of sacubitril/valsartan has been demonstrated to be stronger than that of enalapril [14, 31]. Additionally, sacubitril/valsartan was found to be more effective than enalapril in reducing risks of death and hospitalization due to heart failure [32]. The improved prognosis with sacubitril/valsartan may be directly related to the suppression of aldosterone production that is associated with this medication.
The PAC-lowering effect of sacubitril/valsartan may be due to the mechanism of AT1 receptor inhibition by the ARBs in sacubitril/valsartan. In one study involving angiotensin II-sensitized human adrenocortical cells, however, the addition of sacubitril or valsartan alone did not inhibit aldosterone synthesis, and sacubitril inhibited aldosterone synthesis only when acting in conjunction with an NP [11]. In other words, the mechanism of the aldosterone-lowering effect of sacubitril/valsartan seems to be mediated by ARBs as well as enhancement of NP due to inhibition of neprilysin (Fig. 4).
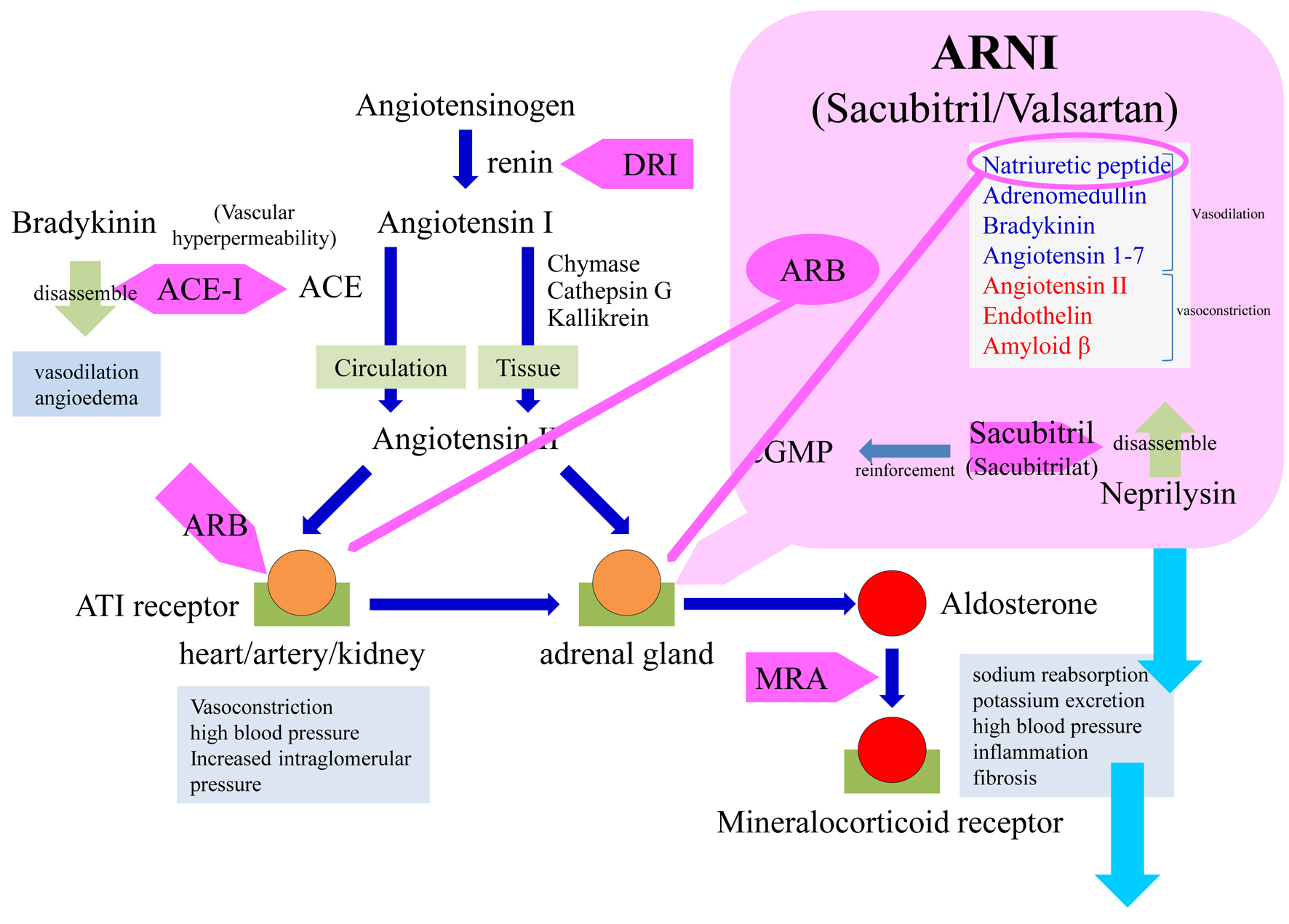 Click for large image |
Figure 4. Schema of the renin-angiotensin-aldosterone system and sacubitril/valsartan. As valsartan blocks angiotensin II type 1 receptors and sacubitril exerts a natriuretic peptide-mediated effect on the adrenal glands, the combination of sacubitril/valsartan directly suppresses aldosterone production and secondarily suppresses the mineralocorticoid receptor-mediated effect. Because the mineralocorticoid receptor is blocked by administration of an MRA, however, positive feedback activates the RAAS and stimulates aldosterone production. ACE: angiotensin-converting enzyme; ACE-I: angiotensin-converting enzyme inhibitor; ARB: angiotensin receptor blocker; ARNI: angiotensin receptor-neprilysin inhibitor; AT: angiotensin; cGMP: cyclic guanosine monophosphate; DRI: direct renin inhibitor; MRA: mineralocorticoid receptor antagonist; RAAS: renin-angiotensin-aldosterone system. |
In a mouse model of type 2 diabetes with aldosterone excess, sacubitril/valsartan increased both renal plasma flow and the glomerular filtration rate and improved tubulointerstitial fibrosis, compared with valsartan or sacubitril alone. The authors of the report suggested that sacubitril and valsartan may act together to protect against aldosterone load-induced damage [33]. In another study that involved patients with HFrEF, despite treatment with angiotensin-converting enzyme inhibitors and beta-blockers, the addition of sacubitril/valsartan was more effective than the addition of MRA [34]. One of the mechanisms of this action may be the inhibitory effect on aldosterone production that is seen with sacubitril/valsartan but absent with MRAs.
Other medications inhibiting aldosterone production
Osilodrostat, an aldosterone synthase inhibitor with similar PAC-lowering effects as sacubitril/valsartan, was developed as an orally available specific inhibitor of aldosterone synthase (CYP11B2).
Oral administration of osilodrostat has been shown to produce a good BP-lowering effect [35], and use of this drug in patients with PA has resulted in significantly reduced PAC [36]. However, osilodrostat also inhibits CYP11B1 and decreases cortisol production, which has the side effect of causing adrenal insufficiency; consequently, this medication has come to be used as a treatment for Cushing disease. Baxdrostat [37], which has greater selectivity for CYP11B2, has demonstrated effectiveness in lowering BP, increasing PRA, and significantly reducing PAC [38]. Clinical trials of baxdrostat are underway [38, 39] and will likely lead to future discussions comparing the pathophysiology underlying reductions in PAC seen with aldosterone synthase inhibitors versus increases in PAC observed with MRAs.
Conclusions
PA involves the pathologically excessive production of aldosterone. In the future, large numbers of patients with PA hopefully will be evaluated to assess how the administration of sacubitril/valsartan affects prognosis and whether decreased PAC can serve as a surrogate marker for prognosis. Given the pathophysiology of PA, more effective treatment may be possible by using sacubitril/valsartan to suppress aldosterone along with MRA therapy to block the action of aldosterone.
Limitation
These three cases were actually diagnosed by first author in other hospital and transferred to the hospital where first author currently work. We changed the patients’ medication as this case reports in our current hospital. They were not referred by a medical practitioner. Therefore, we unfortunately found that detailed patients characteristics had not been recorded in this hospital.
In future, we should conduct statistical analysis on many more cases to confirm this phenomenon. Recently, we have just received permission from the ethical committee to conduct a clinical study to statistically examine changes in plasma aldosterone concentration and plasma renin activity when sacubitril/valsartan is added to patients with primary aldosteronism who are taking MRA.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
We received grant support from Otsuka Holdings for participating in a clinical trial of treating hypertension with an ultrasound renal denervation system.
Informed Consent
Permission to report was obtained from all patients.
Author Contributions
Keisuke Okamura: study design, data collection, statistical analysis, data interpretation, manuscript preparation, and literature search. Masatoshi Matsushima, Yosuke Takamiya, Tetsu Okuda, Hideto Sako, Akihiro Udo, Kenichiro Taniguchi, Shogo Morisaki, Ichiro Imamura, Hidenori Urata, and Shin-ichiro Miura: manuscript preparation and literature search.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
| References | ▴Top |
- Milliez P, Girerd X, Plouin PF, Blacher J, Safar ME, Mourad
JJ. Evidence for an increased rate of cardiovascular events in patients with primary
aldosteronism. J Am Coll Cardiol. 2005;45(8):1243-1248.
doi pubmed - Omura M, Saito J, Yamaguchi K, Kakuta Y, Nishikawa T.
Prospective study on the prevalence of secondary hypertension among hypertensive patients
visiting a general outpatient clinic in Japan. Hypertens Res. 2004;27(3):193-202.
doi pubmed - Takamiya Y, Kitajima K, Kubota K, Matsumoto K, Sumi S,
Okamura K, Mori K, et al. A case of primary aldosteronism who experienced cardiopulmonary
arrest, was resuscitated and cured. J Cardiol Cases. 2014;9(2):63-66.
doi pubmed - Omura M, Sasano H, Saito J, Yamaguchi K, Kakuta Y, Nishikawa
T. Clinical characteristics of aldosterone-producing microadenoma, macroadenoma, and idiopathic
hyperaldosteronism in 93 patients with primary aldosteronism. Hypertens Res.
2006;29(11):883-889.
doi pubmed - Omura M, Sasano H, Fujiwara T, Yamaguchi K, Nishikawa T.
Unique cases of unilateral hyperaldosteronemia due to multiple adrenocortical micronodules,
which can only be detected by selective adrenal venous sampling. Metabolism.
2002;51(3):350-355.
doi pubmed - Kempers MJ, Lenders JW, van Outheusden L, van der Wilt GJ,
Schultze Kool LJ, Hermus AR, Deinum J. Systematic review: diagnostic procedures to differentiate
unilateral from bilateral adrenal abnormality in primary aldosteronism. Ann Intern Med.
2009;151(5):329-337.
doi pubmed - Satoh M, Maruhashi T, Yoshida Y, Shibata H. Systematic
review of the clinical outcomes of mineralocorticoid receptor antagonist treatment versus
adrenalectomy in patients with primary aldosteronism. Hypertens Res. 2019;42(6):817-824.
doi pubmed - Miyake Y, Tanaka K, Nishikawa T, Naruse M, Takayanagi R,
Sasano H, Takeda Y, et al. Prognosis of primary aldosteronism in Japan: results from a
nationwide epidemiological study. Endocr J. 2014;61(1):35-40.
doi pubmed - Hundemer GL, Curhan GC, Yozamp N, Wang M, Vaidya A.
Cardiometabolic outcomes and mortality in medically treated primary aldosteronism: a
retrospective cohort study. Lancet Diabetes Endocrinol. 2018;6(1):51-59.
doi pubmed - Okamura K, Matsushima M, Yamamoto F, Takamiya Y, Okuda T,
Shirai K, Okamura K, et al. A patient with bilateral primary aldosteronism refractory to oral
eplerenone who responded to Esaxerenone with increased renin activity. Am J Case
Rep. 2020;21:e920615.
doi pubmed - Miura SI, Suematsu Y, Matsuo Y, Tomita S, Nakayama A, Goto
M, Arimura T, et al. The angiotensin II type 1 receptor-neprilysin inhibitor LCZ696 blocked
aldosterone synthesis in a human adrenocortical cell line. Hypertens Res.
2016;39(11):758-763.
doi pubmed - Mochel JP, Teng CH, Peyrou M, Giraudel J, Danhof M, Rigel
DF. Sacubitril/valsartan (LCZ696) significantly reduces aldosterone and increases cGMP
circulating levels in a canine model of RAAS activation. Eur J Pharm Sci.
2019;128:103-111.
doi pubmed - Kobalava Z, Kotovskaya Y, Averkov O, Pavlikova E, Moiseev V,
Albrecht D, Chandra P, et al. Pharmacodynamic and pharmacokinetic profiles of
sacubitril/valsartan (LCZ696) in patients with heart failure and reduced ejection fraction.
Cardiovasc Ther. 2016;34(4):191-198.
doi pubmed - Januzzi JL, Butler J, Fombu E, Maisel A, McCague K, Pina IL,
Prescott MF, et al. Rationale and methods of the prospective study of biomarkers, symptom
improvement, and ventricular remodeling during sacubitril/valsartan therapy for heart failure
(PROVE-HF). Am Heart J. 2018;199:130-136.
doi pubmed - Makita K, Nishimoto K, Kiriyama-Kitamoto K, Karashima S,
Seki T, Yasuda M, Matsui S, et al. A novel method: super-selective adrenal venous sampling. J
Vis Exp. 2017;127:e55716.
doi pubmed - Omura M, Saito J, Matsuzawa Y, Nishikawa T. Supper-selective
ACTH-stimulated adrenal vein sampling is necessary for detecting precisely functional state of
various lesions in unilateral and bilateral adrenal disorders, inducing primary aldosteronism
with subclinical Cushing's syndrome. Endocr J. 2011;58(10):919-920.
doi pubmed - Okamura K, Urata H. Simplifying adrenal vein sampling for
cardiologists "In the New Era of Catheter Treatment for Hypertension". Hypertens Res.
2019;42(1):117-119.
doi pubmed - Nishikawa T, Omura M, Satoh F, Shibata H, Takahashi K,
Tamura N, Tanabe A, et al. Guidelines for the diagnosis and treatment of primary
aldosteronism—the Japan Endocrine Society 2009. Endocr J.
2011;58(9):711-721.
doi pubmed - Nishikawa T, Omura M, Kawaguchi M, Takatsu A, Satoh F, Ito
S, Kurihara I, et al. Calibration and evaluation of routine methods by serum certified reference
material for aldosterone measurement in blood. Endocr J. 2016;63(12):1065-1080.
doi pubmed - Ichikawa S, Tajima Y, Sakamaki T, Matsuo H, Kogure M, Hirano
Y, Yagi S, et al. Effect of spironolactone on fluid volumes and adrenal steroids in primary
aldosteronism. Jpn Circ J. 1984;48(11):1184-1196.
doi pubmed - Arai K, Homma T, Morikawa Y, Ubukata N, Tsuruoka H, Aoki K,
Ishikawa H, et al. Pharmacological profile of CS-3150, a novel, highly potent and selective
non-steroidal mineralocorticoid receptor antagonist. Eur J Pharmacol.
2015;761:226-234.
doi pubmed - Sato A, Hayashi K, Naruse M, Saruta T. Effectiveness of
aldosterone blockade in patients with diabetic nephropathy. Hypertension.
2003;41(1):64-68.
doi pubmed - Lucatello B, Benso A, Tabaro I, Capello E, Caprino MP,
Marafetti L, Rossato D, et al. Long-term re-evaluation of primary aldosteronism after medical
treatment reveals high proportion of normal mineralocorticoid secretion.
Eur J Endocrinol. 2013;168(4):525-532.
doi pubmed - Yoneda T, Demura M, Takata H, Kometani M, Karashima S,
Yamagishi M, Takeda Y. Unilateral primary aldosteronism with spontaneous remission after
long-term spironolactone therapy. J Clin Endocrinol Metab. 2012;97(4):1109-1113.
doi pubmed - Yoshida Y, Fujiki R, Kinoshita M, Sada K, Miyamoto S, Ozeki
Y, Mori Y, et al. Importance of dietary salt restriction for patients with primary aldosteronism
during treatment with mineralocorticoid receptor antagonists: The potential importance of
post-treatment plasma renin levels. Hypertens Res. 2023;46(1):100-107.
doi pubmed - Hosoda K, Nakao K, Mukoyama M, Saito Y, Jougasaki M,
Shirakami G, Suga S, et al. Expression of brain natriuretic peptide gene in human heart.
Production in the ventricle. Hypertension. 1991;17(6 Pt 2):1152-1155.
doi pubmed - Yamamoto K, Rakugi H. Angiotensin receptor-neprilysin
inhibitors: Comprehensive review and implications in hypertension treatment. Hypertens Res.
2021;44(10):1239-1250.
doi pubmed - D'Elia E, Iacovoni A, Vaduganathan M, Lorini FL, Perlini S,
Senni M. Neprilysin inhibition in heart failure: mechanisms and substrates beyond modulating
natriuretic peptides. Eur J Heart Fail. 2017;19(6):710-717.
doi pubmed - Munzel T, Kurz S, Holtz J, Busse R, Steinhauer H, Just H,
Drexler H. Neurohormonal inhibition and hemodynamic unloading during prolonged inhibition of ANF
degradation in patients with severe chronic heart failure. Circulation.
1992;86(4):1089-1098.
doi pubmed - Gu J, Noe A, Chandra P, Al-Fayoumi S, Ligueros-Saylan M,
Sarangapani R, Maahs S, et al. Pharmacokinetics and pharmacodynamics of LCZ696, a novel
dual-acting angiotensin receptor-neprilysin inhibitor (ARNi). J Clin Pharmacol.
2010;50(4):401-414.
doi pubmed - Zile MR, O'Meara E, Claggett B, Prescott MF, Solomon SD,
Swedberg K, Packer M, et al. Effects of sacubitril/valsartan on biomarkers of extracellular
matrix regulation in patients with HFrEF. J Am Coll Cardiol. 2019;73(7):795-806.
doi pubmed - McMurray JJ, Packer M, Desai AS, Gong J, Lefkowitz MP,
Rizkala AR, Rouleau JL, et al. Angiotensin-neprilysin inhibition versus enalapril in heart
failure. N Engl J Med. 2014;371(11):993-1004.
doi pubmed - Nishio H, Ishii A, Yamada H, Mori KP, Kato Y, Ohno S, Handa
T, et al. Sacubitril/valsartan ameliorates renal tubulointerstitial injury through increasing
renal plasma flow in a mouse model of type 2 diabetes with aldosterone excess. Nephrol Dial
Transplant. 2023;38(11):2517-2527.
doi pubmed - Escobar C, Barrios V, Manzano L. Switching to
sacubitril/valsartan or adding aldosterone antagonist: which first? ESC Heart Fail.
2019;6(6):1334-1335.
doi pubmed - Calhoun DA, White WB, Krum H, Guo W, Bermann G, Trapani A,
Lefkowitz MP, et al. Effects of a novel aldosterone synthase inhibitor for treatment of primary
hypertension: results of a randomized, double-blind, placebo- and active-controlled phase 2
trial. Circulation. 2011;124(18):1945-1955.
doi pubmed - Amar L, Azizi M, Menard J, Peyrard S, Watson C, Plouin PF.
Aldosterone synthase inhibition with LCI699: a proof-of-concept study in patients with primary
aldosteronism. Hypertension. 2010;56(5):831-838.
doi pubmed - Bogman K, Schwab D, Delporte ML, Palermo G, Amrein K, Mohr
S, De Vera Mudry MC, et al. Preclinical and early clinical profile of a highly selective and
potent oral inhibitor of aldosterone synthase (CYP11B2). Hypertension.
2017;69(1):189-196.
doi pubmed - Freeman MW, Halvorsen YD, Marshall W, Pater M, Isaacsohn J,
Pearce C, Murphy B, et al. Phase 2 trial of Baxdrostat for treatment-resistant hypertension.
N Engl J Med. 2023;388(5):395-405.
doi pubmed - Hayashi T, Shibata H, Kurihara I, Yokota K, Mitsuishi Y,
Ohashi K, Murai-Takeda A, et al. High glucose stimulates mineralocorticoid receptor
transcriptional activity through the protein kinase C beta signaling. Int Heart J.
2017;58(5):794-802.
doi pubmed
This
article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0
International License, which permits unrestricted non-commercial use, distribution, and
reproduction in any medium, provided the original work is properly cited.
Journal
of Clinical Medicine Research is published by Elmer Press Inc.