| Journal of Clinical Medicine Research, ISSN 1918-3003 print, 1918-3011 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Med Res and Elmer Press Inc |
| Journal website https://jocmr.elmerjournals.com |
Original Article
Volume 000, Number 000, March 2025, pages 000-000
Comparative Evaluation of Risk of Death in Mechanically Ventilated Patients With COVID-19 and Influenza: A Population-Based Cohort Study
Lavi Ouda, c, John Garzab
aDivision of Pulmonary and Critical Care Medicine, Department of Internal Medicine, Texas Tech University Health Sciences Center at the Permian Basin, Odessa, TX 79763, USA
bDepartment of Pediatrics, Texas Tech University Health Sciences Center at the Permian Basin, Odessa, TX 79763, USA
cCorresponding Author: Lavi Oud, Division of Pulmonary and Critical Care Medicine, Department of Internal Medicine, Texas Tech University Health Sciences Center at the Permian Basin, Odessa, TX 79763, USA
Manuscript submitted January 24, 2025, accepted March 10, 2025, published online March 17, 2025
Short title: Mortality in COVID-19 vs. Influenza
doi: https://doi.org/10.14740/jocmr6189
| Abstract | ▴Top |
Background: Reports on the comparative mortality among mechanically ventilated patients with coronavirus disease 2019 (COVID-19) and influenza show conflicting findings, but studies focused largely on the early phase of the pandemic, using historical influenza comparators. We sought to examine the population-level comparative mortality among mechanically ventilated patients with COVID-19 during the latter pandemic years using contemporaneous influenza comparators.
Methods: We used a statewide dataset to identify mechanically ventilated hospitalizations aged ≥ 18 years with COVID-19 or influenza in Texas between October 2021 and March 2023. Their comparative short-term mortality (in-hospital death or discharge to hospice) was estimated using overlap propensity score weighting (primary model), entropy balance, and hierarchical logistic models.
Results: Among 22,195 mechanically ventilated hospitalizations, 19,659 (88.6%) had COVID-19 and 2,536 (11.4%) had influenza. Compared to mechanically ventilated hospitalizations with influenza, those with COVID-19 were more commonly racial or ethnic minority (49.3% vs. 48.4%) and had lower mean (standard deviation (SD)) Deyo comorbidity index (2.04 (2.03) vs. 2.53 (1.91)), but higher number of organ dysfunctions (2.60 (1.37) vs. 2.13 (1.27)), respectively. Short-term mortality among mechanically ventilated hospitalizations with COVID-19 and influenza was 49.1% vs. 20.7%. The risk of short-term mortality was attenuated but remained higher among hospitalizations with COVID-19 in the primary model (adjusted risk ratio: 1.24 (95% confidence interval (CI): 1.18 - 1.30); adjusted risk difference 8.8% (95% CI: 6.7 - 10.4)), with consistent findings in alternative models, subgroups, and sensitivity analyses.
Conclusions: Population-level short-term mortality among mechanically ventilated hospitalizations with COVID-19 has been higher than that among those with influenza during the latter years of the pandemic.
Keywords: COVID-19; Influenza; Mechanical ventilation; Mortality
| Introduction | ▴Top |
The coronavirus disease 2019 (COVID-19) pandemic has resulted in a tremendous global morbidity and mortality toll [1]. In the United States (USA), by the time of the official declaration of the end of the public health emergency phase of the pandemic in May 2023 [2], the national mortality toll of COVID-19 infections was over 1 million deaths [3]. Nevertheless, there has been substantial decline in mortality of patients with COVID-19 during the pandemic years [4], including among hospitalized patients [5, 6].
Prior to the COVID-19 pandemic, influenza was the most common cause of hospitalization and mortality due to respiratory viral infections in the USA [7], with annual estimates of hospitalizations and deaths up to 700,000 and 51,000, respectively, during the prepandemic decade [8]. However, there has been dramatic decrease in influenza activity worldwide during the 2020 - 2021 influenza season [9], precluding even national estimates of the number of influenza-related hospitalizations and deaths in the USA during that period [8]. Because both infections share common risk factors and clinical, predominantly respiratory, manifestations [10], numerous studies sought to examine their comparative mortality burden, generally showing higher mortality rates among hospitalized patients with COVID-19 compared to those with influenza [11, 12], including during the latter years of the pandemic [5, 13].
Hospitalized patients with COVID-19 and influenza can present with or subsequently develop severe respiratory failure requiring mechanical ventilation, which has been associated with exceedingly high mortality among the former [14]. Thus, not unexpectedly, mortality among patients with COVID-19 was reported to be driven by those requiring mechanical ventilation [15, 16]. However, the evidence on the comparative outcomes of mechanically ventilated patients with COVID-19 and influenza shows conflicting findings, with COVID-19 patients reported to have higher [17, 18], lower [19, 20], or similar [21, 22] mortality rates compared to those with influenza. Comparisons across these studies and their generalizability are hampered by common monocentric design, small cohort size, limitations of analytical approach and, importantly, by use of historical rather than contemporaneous influenza comparators, as well as predominant focus on the early phase of the pandemic. Thus, these studies may not be representative of the more contemporaneous comparative outcomes of mechanically ventilated patients with COVID-19 vs. influenza, given the substantial decrease in mortality among patients with COVID-19 [4-6], reflecting the interplay between evolving intrinsic viral virulence, growing infection- and vaccine-related population immunity, availability of antiviral and immunomodulatory agents, and advances in the care of severely ill patients over time.
Although COVID-19 is no longer considered a public health emergency in the USA [2], it remains a leading cause of respiratory viral hospitalizations [23], and nearly 77,000 COVID-19-related deaths were reported in 2023 [24]. It is, however, unknown whether the short-term outcomes of mechanically ventilated patients with COVID-19 have become comparable to those with other common respiratory viral infections, such as influenza. These data can inform health policy and resource allocation, clinician decision-making, and serve as benchmarks for preventive and interventional efforts to mitigate the toll of COVID-19 in this population subset. We sought to examine the population-level short-term mortality among mechanically ventilated patients with COVID-19 compared to contemporaneous counterparts with influenza.
| Materials and Methods | ▴Top |
Study design and data sources
This was a retrospective, population-based cohort study. Because we used a publicly available, deidentified dataset, the study was determined to be exempt from formal review by the Texas Tech Health Sciences Center Institutional Review Board. The reporting of the study findings follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines on reporting observational studies in epidemiology [25].
We used the Texas Inpatient Public Use Data File (TIPUDF) to identify the target population. In brief, the TIPUDF is an administrative dataset maintained by the Texas Department of State Health Services [26] and includes inpatient discharge data from state-licensed, non-federal hospitals, and captures approximately 97% of all hospital discharges in the state.
The information on diseases and procedures in the dataset is based on International Classification of Diseases codes. Thus, the TIPUDF does not contain clinical information, such as patients’ clinical condition at the time of initiation of mechanical ventilation, indications for mechanical ventilation, ventilation strategies and settings, or physiological data.
Study population
We have identified hospitalizations aged 18 years or older requiring mechanical ventilation in acute care hospitals with a diagnosis of COVID-19 or influenza between October 1, 2021, and March 31, 2023. Mechanical ventilation was defined by the International Classification of Diseases, 10th Revision (ICD-10) procedure codes 5A1935Z, 5A1945Z, and 5A1955Z. Hospitalizations with COVID-19 were identified by ICD-10 code U071 [17, 27, 28] and those with influenza by ICD-10 codes J09x, J10x, and J11x [22, 27, 29, 30]. We have excluded hospitalizations related to pregnancy (n = 86) and those with diagnosis codes for both COVID-19 and influenza (n = 279). In addition, we excluded hospitalizations transferred from or discharged to another acute care hospital (n = 2,997) because these lacked data on complete hospital course [17, 18].
Outcome
The primary outcome was short-term mortality among mechanically ventilated hospitalizations. Short-term mortality was defined as the combination of in-hospital mortality or discharge to hospice. We selected this composite outcome because in-hospital mortality rates in the USA are known to be influenced by nonclinical variables, including hospital transfer practices [31, 32]. Specifically, discharges to hospice among hospitalized patients have progressively increased in the USA over the past decades and were associated with corresponding decrease in in-hospital mortality across broad categories of patients [33], which shifts attribution of death from index hospitalization to hospice [34]. These data suggest that in-hospital mortality rates can bias estimates across groups [35]. As a result, this composite outcome has been increasingly used in epidemiological studies [36, 37].
Risk-adjustment covariates
Risk-adjustment covariates were selected a priori based on biological and clinical plausibility and existing literature [38-43] and included patients’ sociodemographic variables (age, gender, race/ethnicity, primary health insurance), coexisting conditions (conditions included in the Deyo modification of the Charlson comorbidity index [44, 45], obesity, tobacco use, alcohol use and substance use disorders), measures of illness severity, organ support interventions (hemodialysis and extracorporeal membrane oxygenation (ECMO)), do-not-resuscitate status (ICD-10 code Z66), palliative care (ICD-10 code Z515), and hospitals’ teaching status. In addition, we abstracted the year and calendar quarter of hospitalization. A calendar quarter represents the shortest time period reported in TIPUDF.
We used the Clinical Classifications Software Refined (CCSR) tool [46] to identify ICD-10 codes for obesity (CCSR category END009), tobacco use (category MBD024), alcohol use (category MBD017), and substance use disorders (categories MBD018 - MBD023, MBD025, and MBD028 - MBD033). Severity of illness was characterized using ICD-10 codes for organ dysfunction [47, 48]. ICD-10 procedure codes were used to identify hospitalizations with hemodialysis (5A1D00Z, 5A1D60Z, 5A1D70Z, 5A1D80Z, 5A1D90Z) and ECMO (5A15223, 5A1522F, 5A1522G, 5A1522H).
Statistical analysis
We summarized categorical variables as frequencies and percentages, while continuous variables were reported as mean (standard deviation (SD)). The Chi-square test was used for group comparison involving categorical variables, while t-test was used for comparison of continuous variables.
Because the TIPUDF dataset provides discharge-level, rather than patient-level information, which precludes accounting for repeated admissions, we report the number of hospitalizations as units of analysis, rather than number of patients.
We used three distinct models to adjust for confounding: 1) overlap propensity score weighting (primary model); 2) entropy balance; and 3) hierarchical multivariable logistic regression. The primary model was also used for subgroup analyses and sensitivity analyses.
Overlap propensity score weighting
Overlap weighting is a technique that assigns weight to each patient equal to the probability of being assigned to the opposite exposure group [49]. The method assigns less weight to those with outlier propensity scores (and more weight to those with propensity score close to 0.5). Thus, overlap weights prevent subjects with extreme propensity scores from dominating results and worsen precision, as can occur with inverse probability treatment weighting. Overlap weighting for the two groups will always lead to an exact balance in the means and proportions of all measured covariates leading to an absolute standardized difference of 0 when propensity score is estimated by logistic regression, does not exclude any study participants, and was shown to optimize precision of estimates compared with other types of propensity score-based methods [49]. Thus, overlap weighting has been considered as effective as randomization if no adjustment was needed [50]. We calculated overlap weights using all the covariates listed earlier under risk-adjustment. Because healthcare data are often hierarchical, we have accounted for the spatial (outcomes affected by facility of hospitalization) and temporal (potential outcome differences in groups among hospitalizations during different time periods) clustering of our data in creating overlap weights for individual hospitals and the quarter of hospitalization, as recommended by Li et al [51]. We used weighted logistic regression with robust standard error to estimate the risk of short-term mortality among mechanically ventilated hospitalizations with COVID-19 compared to those with influenza.
Entropy balance
Entropy balancing is a data processing method that assigns scalar weight to each patient, incorporating covariate balance into the weight function, so that covariate distribution in the reweighted data achieves exact balance, capturing the average difference in outcomes across the full population studied [52]. Balance improvement with entropy balancing reduces model dependence for effect estimates. All the covariates listed earlier under risk-adjustment were included in estimating entropy balancing weights. Similar to overlap weighting, we have accounted for the spatial and temporal clustering of our data, using the approach reported by Xu et al [53]. Weighted logistic regression with robust standard error was used to estimate the risk of short-term mortality among mechanically ventilated hospitalizations with COVID-19 compared to those with influenza.
Hierarchical multivariable logistic regression
We fit a hierarchical logistic regression model, adjusted for all the covariates listed in the risk-adjustment section. We nested mechanically ventilated hospitalizations within individual hospitals and treated hospitals as a random effect. Multicollinearity was excluded by examination of variance inflation factors.
The estimates of comparative short-term mortality of mechanically ventilated hospitalizations with COVID-19 vs. those with influenza in the weighted logistic regressions of the overlap propensity score weighting, the entropy balance model, and in the hierarchical multivariable logistic regression model were expressed as adjusted risk ratio (aRR) and adjusted risk difference (aRD), using the approach described by Austin [54]. The aRD represents the absolute difference in short-term mortality, expressed as percentage, between mechanically ventilated hospitalizations with COVID-19 compared to those with influenza. Non-parametric bootstrap 95% confidence intervals (CIs) were determined using 10,000 bootstrap samples and the percentile method, as described by Austin [54].
The State of Texas suppresses gender data of hospitalizations with a diagnosis of infection with the human immunodeficiency virus, and of those with alcohol or drug use to protect patients’ identity. Gender data were missing nonrandomly in 7.8% of hospitalizations in our cohort, precluding imputation of missing values. We used an indicator variable to model gender data for hospitalizations with suppressed gender information. Randomly missing data (1% of health insurance) were modelled as indicator variable.
Subgroup analyses
We performed pre-specified subgroup analyses within the overlap propensity score weighted cohort, including: 1) age; 2) gender; 3) race and ethnicity; 4) Deyo comorbidity index; and 5) the number of organ dysfunctions. Age, gender, race and ethnicity, Deyo comorbidity index, and the number of organ dysfunctions, were excluded from models 1 - 5, respectively.
Sensitivity analyses
We performed four sensitivity analyses to assess the robustness of the primary analysis estimates. First, we examined the comparative outcomes of mechanically ventilated hospitalizations with COVID-19 and influenza using in-hospital mortality as an outcome. Second, we repeated the primary analysis on the subset of mechanically ventilated hospitalizations with COVID-19 and influenza, restricted to the periods from October 1, 2021 to March 31, 2022 and October 1, 2022 to March 31, 2023, approximating the approach of the Centers of Disease Control and Prevention for the definition of the influenza season in the USA [55, 56], as influenza-related mortality would be expected to be higher during this period. Third, because COVID-19 mortality was reported to decrease over time, we examined the comparative short-term mortality of the subsets of mechanically ventilated hospitalizations with COVID-19 and influenza during the third and fourth quarters of 2022 (from July 1 to September 30 and October 1 to December 31, respectively), as well as last study quarter (from January 1 to March 31, 2023), with the latter representing the latest time period in our cohort prior to the end of the public health emergency phase of the pandemic in the USA [2], and thus potentially reflecting the smallest mortality gap between the examined groups.
Finally, to account for unmeasured confounding, we computed E-values [57] to assess the magnitude of a potential unmeasured confounder required to nullify the estimates of the primary model of the comparative short-term mortality of mechanically ventilated hospitalizations with COVID-19 and influenza. The E-value represents the minimum strength of association, on a risk ratio scale, that an unmeasured confounder would need with both the exposure and outcome, conditioned on the measured covariates, to fully explain a specific association [57]. A large E-value implies that a considerable unmeasured confounding would be needed to explain away the effect estimate. We report the E-values for the point estimate and the lower bound of the 95% CI for our primary model.
Data management was performed using Microsoft Excel (Microsoft, Redmond, Washington) and statistical analyses were performed with R 4.0.5 (R Foundation for Statistical Computing, Vienna, Austria). A two-sided P value < 0.05 was considered statistically significant.
| Results | ▴Top |
Cohort characteristics
There were 22,195 mechanically ventilated hospitalizations during the study period, of which 19,659 (88.6%) had COVID-19 and 2,536 (11.4%) had a diagnosis of influenza.
The characteristics of the mechanically ventilated hospitalizations with COVID-19 and influenza are detailed in Table 1. Compared to those with influenza, mechanically ventilated hospitalizations with COVID-19 had similar frequency of those with older age (aged ≥ 65 years in 54.9% vs. 54.8%), had higher frequency of racial and ethnic minorities (49.3% vs. 48.4%), but with lower mean (SD) burden of comorbidities (Deyo comorbidity index 2.04 (2.03) vs. 2.53 (1.91)). Among individual comorbid conditions, mechanically ventilated hospitalizations with COVID-19 had higher frequency of diabetes (42.4% vs. 39.0%) and obesity (33.6% vs. 30.8%), but lower frequency of congestive heart failure (33.8% vs. 48.5%) and lung disease (28.5% vs. 57.7%) compared to those with influenza. The mean (SD) number of organ dysfunctions was higher among mechanically ventilated hospitalizations with COVID-19 than among those with influenza (2.60 (1.37) vs. 2.13 (1.27)). The frequency of do-not-resuscitate state and use of palliative care was markedly higher among mechanically ventilated hospitalizations with COVID-19 than among those with influenza (33.1% vs. 19.0% and 22.1% vs. 12.5%, respectively).
 Click to view | Table 1. The Characteristics and Outcomes of Mechanically Ventilated Hospitalizations With COVID-19 and Influenza |
Discharge to home occurred in 29.7% of mechanically ventilated hospitalizations with COVID-19 and in 57.5% of those with influenza, while rates of discharge to a post-acute care facility were similar (19.8% vs. 19.2%, respectively).
Mortality among mechanically ventilated hospitalizations with COVID-19 and influenza
Crude short-term mortality among mechanically ventilated hospitalizations with COVID-19 and influenza was 49.1% vs. 20.7%, respectively. The corresponding hospital mortality was 40.5% among those with COVID-19 and 13.5% among those with influenza.
Estimates of the risk of short-term mortality among mechanically ventilated hospitalizations with COVID-19 compared to those with influenza are detailed in Table 2. Details of the weighted cohorts following overlap weighting and entropy balancing are provided here (Supplementary Materials 1, 2, jocmr.elmerjournals.com), respectively, and the standardized differences between mechanically ventilated hospitalizations with COVID-19 and influenza in the unweighted vs. overlap weighted and entropy balanced cohorts are presented here (Supplementary Materials 3, 4, jocmr.elmerjournals.com), respectively. After applying overlap propensity score weighting, the comparative risk of short-term mortality among mechanically ventilated hospitalizations with COVID-19 vs. those with influenza was attenuated but remained higher among the former (aRR 1.24 (95% CI: 1.18 - 1.30); aRD 8.8% (95% CI: 6.7 - 10.4)). The estimates of the risk of short-term mortality among mechanically ventilated hospitalizations with COVID-19 vs. those with influenza in the alternative models were consistent with those in the primary model. Similarly, the findings in subgroup analyses (Fig. 1) were consistent with those in the primary model, but the higher risk of short-term mortality on point estimates among those aged 18 - 44 years and in the other group of race and ethnicity categories with COVID-19 did not reach statistical significance.
 Click to view | Table 2. Adjusted Analyses of the Comparative Short-Term Mortalitya Among Mechanically Ventilated Hospitalizations With COVID-19 Versus Influenza |
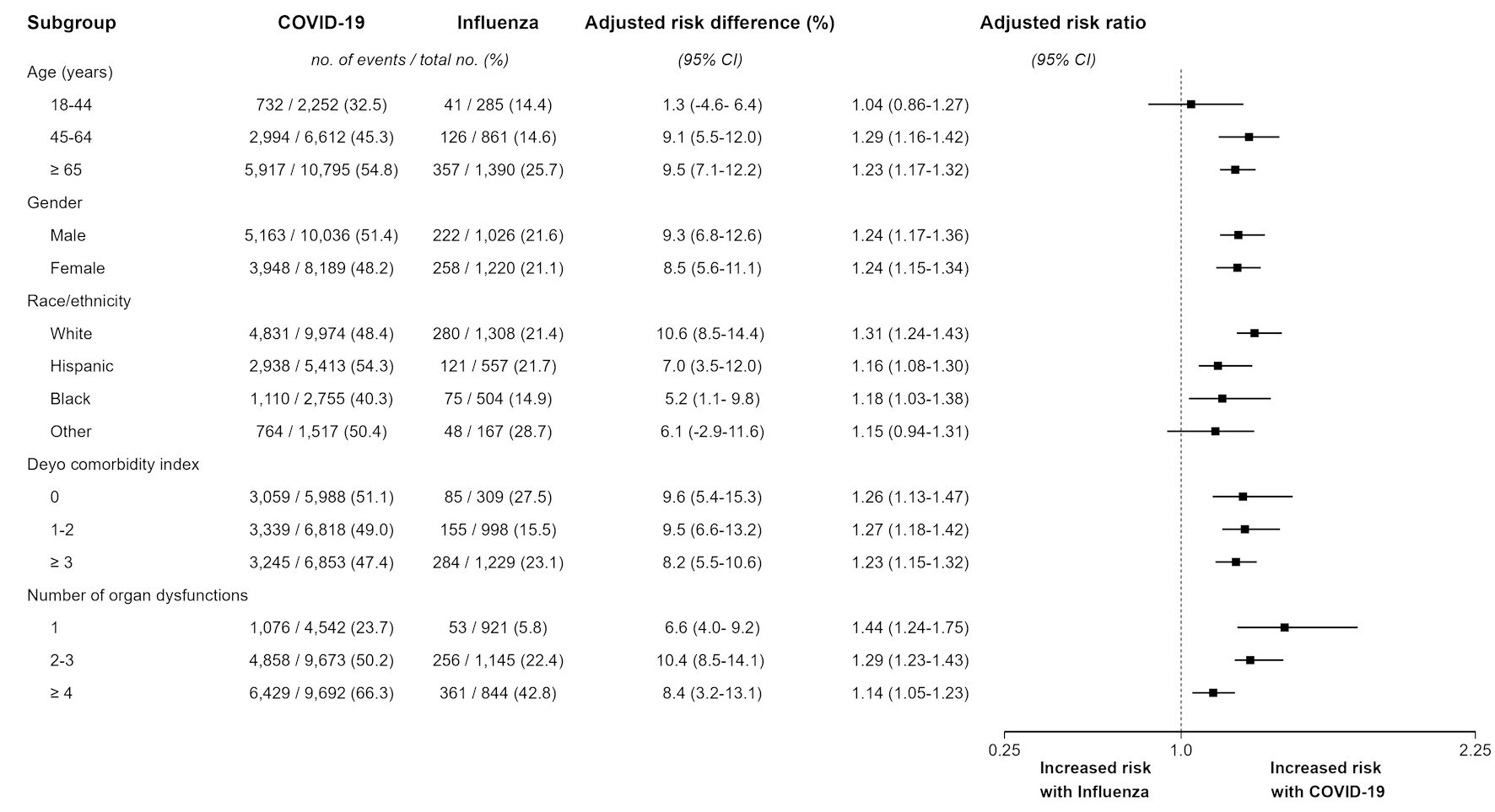 Click for large image | Figure 1. Subgroup analysis of comparative short-term mortality among mechanically ventilated hospitalizations with COVID-19 and influenza. Analyses were conducted using overlap propensity score weighting. Adjusted risk ratio represents the risk of short-term mortality among mechanically ventilated hospitalizations with COVID-19 compared to those with influenza. Adjusted risk difference represents the absolute difference in short-term mortality among mechanically ventilated hospitalizations with COVID-19 compared to those with influenza, expressed as percentage. Non-parametric bootstrap 95% confidence intervals (CIs) were determined using 10,000 bootstrap samples and the percentile method. The width of the CIs was not adjusted for multiplicity and should not be used to infer definite effects. COVID-19: coronavirus disease 2019. |
The findings on sensitivity analyses are detailed here (Supplementary Material 5, jocmr.elmerjournals.com), with crude mortality being markedly higher among mechanically ventilated hospitalizations with COVID-19 compared to those with influenza for all analyzed strata. These analyses were consistent with the findings of the primary model, though the estimates for aRR and aRD for the third quarter of 2022, while higher for COVID-19 vs. influenza, did not reach statistical significance due to a very small number of mechanically ventilated influenza hospitalizations during the off-season period. The risk of short-term mortality remained higher among mechanically ventilated hospitalizations with COVID-19 compared to those with influenza during the last quarter of the study (aRR: 1.19 (95% CI: 1.07 - 1.34); aRD: 5.4% (95% CI: 2.3 - 9.0)). The E-values for the point estimate and the lower bound 95% CI of risk ratio of short-term mortality in the primary model were 1.79 and 1.64, respectively.
| Discussion | ▴Top |
Principal findings
In this population-based study, a diagnosis of COVID-19 was associated with higher risk of death among adult mechanically ventilated hospitalizations compared to those with influenza. We found that the relative risk of short-term mortality was 24% higher among those with COVID-19, corresponding to nearly 9% higher absolute risk of death on adjusted analyses. The higher risk of short-term mortality among mechanically ventilated hospitalizations with COVID-19 was consistent on alternative modelling, subgroups analyses, and remained robust on sensitivity analyses. To our knowledge, this study represents the first population-level examination of comparative short-term mortality among mechanically ventilated hospitalizations with COVID-19 and those with contemporaneously managed influenza during the later years of the pandemic.
Comparison with other studies
Our findings of higher risk of death among mechanically ventilated hospitalizations with COVID-19 compared to those with influenza are concordant with those in several prior reports [17, 18, 27, 29, 58]. The largest among these works was a population-based study from France by Naouri et al, which compared mechanically ventilated patients with COVID-19 from March to June 2020 to those with influenza, hospitalized between 2014 and 2019 [18]. The other studies showing similar findings examined patients with COVID-19 in France [27, 29], Germany [17], and Mexico [58], mostly during the first few months of the pandemic in 2020 [17, 18, 29, 58] or through April 2021 [27], and all have used historical influenza comparators. Although influenza hospitalizations occur year-round, beyond the influenza season, the investigators likely chose historical influenza comparators in part due to lack of overlap between the study period of their COVID-19 patients and seasonal influenza and the substantial decrease in influenza activity during the 2020 - 2021 influenza season [9], which could have led to insufficient number of influenza patients for comparative estimates.
However, despite similar findings, our study cannot be directly compared to the above reports. The outcomes of patients with COVID-19 during the early phase of the pandemic when patients’ mortality was at its highest do not reflect the substantial decrease in their risk of death in latter pandemic years [4, 5], thus resulting in higher gap in mortality when compared to those with influenza. Correspondingly, the use of historical, rather than contemporaneous, influenza comparators may have led to further overestimation of the mortality difference between them and those with COVID-19 in these studies. This likely impact of the use of historical instead of contemporaneous influenza comparators stems from not accounting for the reported increased mortality among non-COVID-19 hospitalizations, especially during the early period of the pandemic [59], which may have similarly affected mechanically ventilated patients with influenza. These worsened outcomes among hospitalized non-COVID-19 patients may reflect in turn both pre-hospital and hospital-level pandemic-related changes. The COVID-19 pandemic has been associated with substantial increase in the severity of illness [60] and, specifically, illness requiring critical care [61], among non-COVID-19 patients presenting to the emergency department, compared to the prepandemic period, which could have increased patients’ risk of death. These pandemic-associated changes in baseline illness severity may have been driven by reported prevalent delays in health care, related to avoidance of medical encounters due to patients’ concerns of exposure to COVID-19 [62, 63], but also due to increased difficulties getting an appointment [62, 64], or access to care location [64]. Moreover, once acute health crises have developed, requiring emergent hospital care, pandemic-related strains on emergency medical services have resulted in delayed on-site care [65] and in arrival to the emergency department [66]. At the hospital level, pandemic-related strains, including those on critical care services [67], have been associated with delays in intensive care unit (ICU) admission [68] and have likely affected adversely time-sensitive and other processes of care. Together, these pandemic-related adverse prognostic changes in non-COVID hospitalizations would be expected to result in higher mortality among mechanically ventilated patients with influenza managed during the pandemic period compared to prepandemic years, and thus could have affected estimates of comparative outcomes with patients with COVID-19.
In contrast to our results, several studies of cohorts from Europe [19, 21, 69-72] and Asia [20, 22, 73] reported either similar mortality rates among mechanically ventilated patients with COVID-19 and influenza [21, 22, 69-73] or lower mortality rates among the former [19, 20]. The findings in these studies, which were generally conducted during an earlier phase of the pandemic, may reflect in part small cohorts with monocentric design [19, 20, 22, 70, 72, 73], and lack of adjustment for confounders [19, 20, 69, 71-73]. In addition, the observed similar or lower mortality among mechanically ventilated patients with COVID-19 compared to those with influenza may reflect the reported over seven-fold across-country variation in the risk of death among mechanically ventilated patients with COVID [14]. Thus, in several studies describing lower or similar risk of death among mechanically ventilated patients with COVID-19 vs. influenza, 28-day mortality among the former was as low as 8.9% [19] to 10.7% [70]. On the other hand, the influenza comparators in one study reflected an epidemic in the year prior to onset of the COVID-19 pandemic, which was considered by the authors to be as severe as that among the latter [72], while several studies included influenza hospitalizations that took place during the 2009 H1N1 influenza pandemic [19, 21, 70]. The latter influenza comparators may not be representative of the mortality outcomes of mechanically ventilated patients with influenza managed during other periods and would have increased overall mortality in the influenza group. There have not been, to our knowledge, studies restricting examination of comparative outcomes of mechanically ventilated patients with COVID-19 to those of contemporaneously managed comparators with influenza during the latter years of the pandemic and showing either similar or lower short-term mortality among the former.
Implications of study findings
Our study shows that despite the substantial decrease in mortality of patients infected with COVID-19 over the pandemic years, its lethality among mechanically ventilated patients remained substantially higher than among those with influenza hospitalized contemporaneously during the latter years of the pandemic. More soberingly, we show that even during the first quarter of 2023, shortly prior to the end of the public health emergency phase of the COVID-19 pandemic in the USA, short-term mortality remained higher among mechanically ventilated hospitalizations with COVID-19 compared to those with influenza. This persistent prognostic gap serves as a reminder that despite the aforementioned growth in population immunity against COVID-19 and major advances in the care of affected patients, COVID-19 cannot be considered another “bad flu” among these critically ill patients.
Presently, although the pandemic-related hospital strain has decreased compared to the early pandemic years, the pandemic’s adverse effects on the healthcare system linger. Thus, a recent study showed that following the end of the public health emergency phase of the pandemic in May 2023 [2], the USA has achieved in 2024 a new hospital occupancy steady state, 11% higher than the prepandemic period (2009 - 2016), driven by a 16% decrease in staffed hospital beds, with hospital occupancy projected to reach a critical level by 2032 [6]. In addition, COVID-19 remains a major cause of respiratory viral hospitalizations in the USA in 2024 [23], while the uptake of the COVID-19 vaccine among adults has decreased in early 2025 to 21.6% (compared to 42.9% who received a 2024 - 2025 influenza vaccine) [74]. Thus, the earlier widespread population immunity against COVID-19 may decrease in the coming years, risking increase in COVID-19-related lethality. Together, the aforementioned evolving trends suggest that COVID-19 remains a clinically relevant health burden, whose lethality may continue to exceed that of influenza.
Further investigations are needed to elucidate the patient-, pathogen-, and health system-related factors underlying the persistent higher lethality of COVID-19 in severely ill patients to inform preventive measures against deterioration of infected patients, development of novel antiviral agents, and refine care of the critically ill.
Strengths and limitations of study
Our study has relevant strengths and limitations. In terms of strengths, our study leverages a population-based, high-quality administrative dataset for the most contemporary estimates of the comparative short-term mortality of consecutive mechanically ventilated hospitalizations with COVID-19 and contemporaneously managed, rather than historical, influenza comparators from a state with a diverse large population of over 30 million. This approach allowed the transcending of local variation in case mix and practice patterns. We adhered closely to reporting guidelines and used multiple statistical methods to limit confounding and enhance the trustworthiness of our estimates of comparative outcomes.
However, population-level analyses using administrative health databases have important limitations, in addition to those noted earlier. The use of ICD codes could have resulted in group misclassification. Although our ICD coding approach has been used by other investigators and was validated, misclassifications could have occurred between hospitalizations with COVID-19 and influenza. However, such potential misclassification would have blurred the difference in mortality between groups. In that case, our findings would represent an underestimate of the true difference in risk of death between mechanically ventilated hospitalizations with COVID-19 and those with influenza.
Our dataset did not include information of patients’ vaccination status, pre-hospitalization and in-hospital pharmacological therapy, data for derivation of prognostic scales (such as Acute Physiology and Chronic Health Evaluation), and processes of care. In addition, our most granular time-based model adjustments were restricted to calendar quarters of hospitalization, rather than dates or weeks, precluding adequate approximation to corresponding evolvement of COVID-19 variants. These variants varied in timing across states, and the reported periods of dominance of a specific variant in the USA notably varied across reports [5, 75, 76]. Further, variation over time in pandemic-induced hospital strain, shown to increase mortality of both COVID-19 and non-COVID critically ill patients during COVID-19 surge conditions [77, 78], could have led to higher mortality among mechanically ventilated patients with both COVID-19 and influenza during high-strain periods and possibly lower mortality for both when hospital strain was lower. However, the resultant strain-related impact on their comparative mortality could not be estimated in our data. Thus, although our adjustment for time-based clustering may have addressed indirectly the variation over time in pandemic-induced hospital strain during the study period, our dataset did not include metrics of hospital strain, precluding credible inferences on its direct impact of our estimates. However, our sensitivity analysis for the last quarter of the study period, which preceded the end of public health emergency in the USA [2] and thus was likely associated with the lowest hospital strain, showed persistence of higher mortality among mechanically ventilated patients with COVID-19 compared to their contemporaneous influenza counterparts. Nevertheless, we cannot exclude residual confounding in our models. However, the E-value for our primary model indicates that the point estimate could be nullified only by an unmeasured confounder with a risk ratio of at least 1.79 for both COVID-19 vs. influenza and for short-term mortality.
Last, our findings may not be generalizable to other states in the USA or to other health systems and geographical regions with different resources and policies.
Conclusions
In this population-based cohort study, short-term mortality among mechanically ventilated hospitalizations with COVID-19 was higher than that among contemporaneously managed influenza comparators during the later years of the pandemic, including in the period preceding the end of the resultant public health emergency in the USA. Additional studies are needed to examine the mechanisms underlying the persistent higher lethality of COVID-19 in severely ill patients, to inform efforts to mitigate its burden in this population.
| Supplementary Material | ▴Top |
Suppl 1. The overlap propensity score weighted characteristics of mechanically ventilated hospitalizations with COVID-19 and influenza.
Suppl 2. The entropy balancing weighted characteristics of mechanically ventilated hospitalizations with COVID-19 and influenza.
Suppl 3. Standardized differences between mechanically ventilated hospitalizations with COVID-19 and influenza for the variables used in the overlap propensity score derivation in the unadjusted and propensity-score weighted cohorts.
Suppl 4. Standardized differences between mechanically ventilated hospitalizations with COVID-19 and influenza for the variables used in the entropy balancing weight derivation in the unadjusted and entropy balance-weighted cohorts.
Suppl 5. Sensitivity analyses of the comparative mortality among mechanically ventilated hospitalizations with COVID-19 vs. influenza.
Acknowledgments
None to declare.
Financial Disclosure
The author received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
Author Contributions
LO: conceptualization, project administration, supervision, and writing - original draft. LO, JG: data curation, investigation, methodology, resources, software, formal analysis, validation, visualization, writing- review and editing. Both authors have read and approved the final manuscript.
Data Availability
The datasets generated and/or analyzed during the current study are available in the TIPUDF repository, at: http://www.dshs.state.tx.us/thcic/hospitals/Inpatientpudf.shtm.
Abbreviations
aRD: adjusted risk difference; aRR: adjusted risk ratio; CCSR: Clinical Classifications Software Refined; COVID-19: coronavirus disease 2019; ICU: intensive care unit; ICD-10: International Classification of Diseases, 10th Revision; TIPUDF: Texas Inpatient Public Use Data File; USA: United States
| References | ▴Top |
- WHO COVID-19 Dashboard. World Health organization. Data. Available from: https://data.who.int/dashboards/covid19/deaths?n=o.
- COVID-1 Public Health Emergency. U.S. Department of Health and Human Services. Coronavirus. Available from: https://www.hhs.gov/coronavirus/covid-19-public-health-emergency/index.html.
- COVID Data Tracker. Centers for Disease Control and Prevention. Available from: https://covid.cdc.gov/covid-data-tracker/#trends_totaldeaths_select_00.
- Horita N, Fukumoto T. Global case fatality rate from COVID-19 has decreased by 96.8% during 2.5 years of the pandemic. J Med Virol. 2023;95(1):e28231.
doi pubmed - Kojima N, Taylor CA, Tenforde MW, Ujamaa D, O'Halloran A, Patel K, Chai SJ, et al. Clinical outcomes of US adults hospitalized for COVID-19 and influenza in the respiratory virus hospitalization surveillance network, October 2021-September 2022. Open Forum Infect Dis. 2024;11(1):ofad702.
doi pubmed - Mukherjee V, Postelnicu R, Parker C, Rivers PS, Anesi GL, Andrews A, Ables E, et al. COVID-19 across pandemic variant periods: the severe acute respiratory infection-preparedness (SARI-PREP) study. Crit Care Explor. 2024;6(7):e1122.
doi pubmed - Nasrullah A, Gangu K, Garg I, Javed A, Shuja H, Chourasia P, Shekhar R, et al. Trends in hospitalization and mortality for influenza and other respiratory viruses during the COVID-19 pandemic in the United States. Vaccines (Basel). 2023;11(2):412.
doi pubmed - Flu disease burden: past seasons. Centers for Disease Control and Prevention. Available from: https://www.cdc.gov/flu-burden/php/data-vis/past-seasons.html.
- Bonacina F, Boelle PY, Colizza V, Lopez O, Thomas M, Poletto C. Global patterns and drivers of influenza decline during the COVID-19 pandemic. Int J Infect Dis. 2023;128:132-139.
doi pubmed - Solomon DA, Sherman AC, Kanjilal S. Influenza in the COVID-19 era. JAMA. 2020;324(13):1342-1343.
doi pubmed - Xie Y, Bowe B, Maddukuri G, Al-Aly Z. Comparative evaluation of clinical manifestations and risk of death in patients admitted to hospital with covid-19 and seasonal influenza: cohort study. BMJ. 2020;371:m4677.
doi pubmed - Sim JK, Lee HS, Yang J, Gwack J, Kim BI, Cha JO, Min KH, et al. Comparative analysis of clinical outcomes using propensity score matching: coronavirus disease 2019 vs. seasonal influenza in Korea. J Korean Med Sci. 2024;39(14):e128.
doi pubmed - Xie Y, Choi T, Al-Aly Z. Risk of death in patients hospitalized for COVID-19 vs seasonal influenza in Fall-Winter 2022-2023. JAMA. 2023;329(19):1697-1699.
doi pubmed - Chandel A, Leazer S, Alcover KC, Farley J, Berk J, Jayne C, McNutt R, et al. Intensive care and organ support related mortality in patients with COVID-19: a systematic review and meta-analysis. Crit Care Explor. 2023;5(3):e0876.
doi pubmed - Nada KM, Hsu ES, Seashore J, Zaidan M, Nishi SP, Duarte A, Sharma G. Determining cause of death during coronavirus disease 2019 pandemic. Crit Care Explor. 2021;3(4):e0419.
doi pubmed - Ketcham SW, Bolig TC, Molling DJ, Sjoding MW, Flanders SA, Prescott HC. Causes and Circumstances of Death among Patients Hospitalized with COVID-19: A Retrospective Cohort Study. Ann Am Thorac Soc. 2021;18(6):1076-1079.
doi pubmed - Ludwig M, Jacob J, Basedow F, Andersohn F, Walker J. Clinical outcomes and characteristics of patients hospitalized for Influenza or COVID-19 in Germany. Int J Infect Dis. 2021;103:316-322.
doi pubmed - Naouri D, Pham T, Dres M, Vuagnat A, Beduneau G, Mercat A, Combes A, et al. Differences in clinical characteristics and outcomes between COVID-19 and influenza in critically ill adult patients: A national database study. J Infect. 2023;87(2):120-127.
doi pubmed - Chung E, Leem AY, Chung KS, Kang YA, Park MS, Kim YS, Jang HJ, et al. Differences of respiratory mechanics in mechanical ventilation of acute respiratory distress syndrome between patients with COVID-19 and Influenza A. Respir Res. 2024;25(1):112.
doi pubmed - Maamar A, Liard C, Doucet W, Reizine F, Painvin B, Delamaire F, Coirier V, et al. Acquired agitation in acute respiratory distress syndrome with COVID-19 compared to influenza patients: a propensity score matching observational study. Virol J. 2022;19(1):145.
doi pubmed - Maamar A, Guillot P, Joussellin V, Delamaire F, Painvin B, Bichon A, de la Jartre OB, et al. Moderate-to-severe ARDS: COVID-19 patients compared to influenza patients for ventilator parameters and mortality. ERJ Open Res. 2023;9(2):00554-2022.
doi pubmed - Taniguchi Y, Kuno T, Komiyama J, Adomi M, Suzuki T, Abe T, Ishimaru M, et al. Comparison of patient characteristics and in-hospital mortality between patients with COVID-19 in 2020 and those with influenza in 2017-2020: a multicenter, retrospective cohort study in Japan. Lancet Reg Health West Pac. 2022;20:100365.
doi pubmed - Respiratory virus hospitalization surveillance network (RESP-NET). Centers for Disease Control and Prevention. Available from: https://www.cdc.gov/resp-net/dashboard/index.html.
- Ahmad FB, Cisewski JA, Anderson RN. Mortality in the United States - provisional data, 2023. MMWR Morb Mortal Wkly Rep. 2024;73(31):677-681.
doi pubmed - von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP, Initiative S. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. PLoS Med. 2007;4(10):e296.
doi pubmed - Texas inpatient public use data file. Texas Department of State Health Services, Center for Health Statistics, Austin, Texas. 2018. Available from: http://www.dshs.state.tx.us/thcic/hospitals/Inpatientpudf.shtm.
- Marois C, Nedelec T, Pelle J, Rozes A, Durrleman S, Dufouil C, Demoule A. Comparison of clinical profiles and mortality outcomes between influenza and COVID-19 patients invasively ventilated in the ICU: a retrospective study from all paris public hospitals from 2016 to 2021. Crit Care Explor. 2022;4(7):e0737.
doi pubmed - Bosch NA, Law AC, Peterson D, Walkey AJ. Validation of the international classification of diseases code for COVID-19 among critically ill patients. Ann Am Thorac Soc. 2022;19(5):861-863.
doi pubmed - Piroth L, Cottenet J, Mariet AS, Bonniaud P, Blot M, Tubert-Bitter P, Quantin C. Comparison of the characteristics, morbidity, and mortality of COVID-19 and seasonal influenza: a nationwide, population-based retrospective cohort study. Lancet Respir Med. 2021;9(3):251-259.
doi pubmed - Benack K, Nyandege A, Nonnenmacher E, Jan S, Setoguchi S, Gerhard T, Strom BL, et al. Validity of ICD-10-based algorithms to identify patients with influenza in inpatient and outpatient settings. Pharmacoepidemiol Drug Saf. 2024;33(4):e5788.
doi pubmed - Vasilevskis EE, Kuzniewicz MW, Dean ML, Clay T, Vittinghoff E, Rennie DJ, Dudley RA. Relationship between discharge practices and intensive care unit in-hospital mortality performance: evidence of a discharge bias. Med Care. 2009;47(7):803-812.
doi pubmed - Reineck LA, Pike F, Le TQ, Cicero BD, Iwashyna TJ, Kahn JM. Hospital factors associated with discharge bias in ICU performance measurement. Crit Care Med. 2014;42(5):1055-1064.
doi pubmed - Schorr CA, Angelo M, John G, LeCompte K, Dellinger RP. The association of increasing hospice use with decreasing hospital mortality: an analysis of the national inpatient sample. J Healthc Manag. 2020;65(2):107-120.
doi pubmed - Kozar RA, Holcomb JB, Xiong W, Nathens AB. Are all deaths recorded equally? The impact of hospice care on risk-adjusted mortality. J Trauma Acute Care Surg. 2014;76(3):634-639; discussion 639-641.
doi pubmed - Law AC, Bosch NA, Song Y, Tale A, Lasser KE, Walkey AJ. In-hospital vs 30-day sepsis mortality at US safety-net and non-safety-net hospitals. JAMA Netw Open. 2024;7(5):e2412873.
doi pubmed - Asch DA, Islam MN, Sheils NE, Chen Y, Doshi JA, Buresh J, Werner RM. Patient and hospital factors associated with differences in mortality rates among black and white US medicare beneficiaries hospitalized with COVID-19 infection. JAMA Netw Open. 2021;4(6):e2112842.
doi pubmed - Alrawashdeh M, Klompas M, Simpson SQ, Kadri SS, Poland R, Guy JS, Perlin JB, et al. Prevalence and outcomes of previously healthy adults among patients hospitalized with community-onset sepsis. Chest. 2022;162(1):101-110.
doi pubmed - Rosenthal N, Cao Z, Gundrum J, Sianis J, Safo S. Risk factors associated with in-hospital mortality in a US national sample of patients with COVID-19. JAMA Netw Open. 2020;3(12):e2029058.
doi pubmed - Bhaskaran K, Bacon S, Evans SJ, Bates CJ, Rentsch CT, MacKenna B, Tomlinson L, et al. Factors associated with deaths due to COVID-19 versus other causes: population-based cohort analysis of UK primary care data and linked national death registrations within the OpenSAFELY platform. Lancet Reg Health Eur. 2021;6:100109.
doi pubmed - Hua MJ, Feinglass J. Variations in COVID-19 hospital mortality by patient race/ethnicity and hospital type in Illinois. J Racial Ethn Health Disparities. 2023;10(2):911-919.
doi pubmed - Kaufmann M, Perren A, Cerutti B, Dysli C, Rothen HU, Swiss Society of Intensive Care Medicine. Severity-adjusted ICU mortality only tells half the truth-the impact of treatment limitation in a nationwide database. Crit Care Med. 2020;48(12):e1242-e1250.
doi pubmed - Fuchs L, Anstey M, Feng M, Toledano R, Kogan S, Howell MD, Clardy P, et al. Quantifying the mortality impact of do-not-resuscitate orders in the ICU. Crit Care Med. 2017;45(6):1019-1027.
doi pubmed - Lee YJ, Yoo JW, Hua L, Kim PC, Kim SJ, Shen JJ. Ten-year trends of palliative care utilization associated with multiple sclerosis patients in the United States from 2005 to 2014. J Clin Neurosci. 2018;58:13-19.
doi pubmed - Quan H, Sundararajan V, Halfon P, Fong A, Burnand B, Luthi JC, Saunders LD, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130-1139.
doi pubmed - Quan H, Li B, Couris CM, Fushimi K, Graham P, Hider P, Januel JM, et al. Updating and validating the Charlson comorbidity index and score for risk adjustment in hospital discharge abstracts using data from 6 countries. Am J Epidemiol. 2011;173(6):676-682.
doi pubmed - Clinical Classification Software Refined (CCSR) for ICD-10-CM diagnoses. Health Care Cost & Utilization Project, User Support. Agency for Healthcare Research and Quality. Available from: https://hcup-us.ahrq.gov/toolssoftware/ccsr/dxccsr.jsp.
- Martin GS, Mannino DM, Eaton S, Moss M. The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med. 2003;348(16):1546-1554.
doi pubmed - Bosch NA, Law AC, Rucci JM, Peterson D, Walkey AJ. Predictive validity of the sequential organ failure assessment score versus claims-based scores among critically ill patients. Ann Am Thorac Soc. 2022;19(6):1072-1076.
doi pubmed - Li F, Morgan KL, Zaslavsky AM. Balancing covariates via propensity score weighting. J Am Stat Assoc. 2018;113(521):390-400.
- Li F, Thomas LE, Li F. Addressing extreme propensity scores via the overlap weights. Am J Epidemiol. 2019;188(1):250-257.
doi pubmed - Li F, Zaslavsky AM, Landrum MB. Propensity score weighting with multilevel data. Stat Med. 2013;32(19):3373-3387.
doi pubmed - Hainmueller J. Entropy balancing for causal effects: a multivariate reweighting method to produce balanced samples in observational studies. Political Analysis. 2012;20:25-46.
- Xu Y, Yang E. Hierarchically regularized entropy balancing. Political Analysis. 2023;31:457-464.
- Austin PC. Absolute risk reductions, relative risks, relative risk reductions, and numbers needed to treat can be obtained from a logistic regression model. J Clin Epidemiol. 2010;63(1):2-6.
doi pubmed - Flu Season. Centers for Disease Control and Prevention. Influenza (Flu). Available from: https://www.cdc.gov/flu/about/season/index.html.
- Tenforde MW, Noah KP, O'Halloran AC, Kirley PD, Hoover C, Alden NB, Armistead I, et al. Timing of influenza antiviral therapy and risk of death in adults hospitalized with influenza-associated pneumonia, influenza hospitalization surveillance network (FluSurv-NET), 2012-2019. Clin Infect Dis. 2025;80(2):461-468.
doi pubmed - VanderWeele TJ, Ding P. Sensitivity Analysis in Observational Research: Introducing the E-Value. Ann Intern Med. 2017;167(4):268-274.
doi pubmed - Hernandez-Cardenas C, Lugo-Goytia G, Hernandez-Garcia D, Perez-Padilla R. Comparison of the clinical characteristics and mortality in acute respiratory distress syndrome due to COVID-19 versus due to Influenza A-H1N1pdm09. Med Intensiva (Engl Ed). 2022;46(6):345-347.
doi pubmed - Zampieri FG, Bastos LSL, Soares M, Salluh JI, Bozza FA. The association of the COVID-19 pandemic and short-term outcomes of non-COVID-19 critically ill patients: an observational cohort study in Brazilian ICUs. Intensive Care Med. 2021;47(12):1440-1449.
doi pubmed - Chowdhury N, Eurick-Bering K, Hjaige M, Kenerson R, Revere TA, Reece RJ. The impact of COVID-19 on patient presentations to the emergency department. Cureus. 2022;14(6):e26100.
doi pubmed - Oskvarek JJ, Zocchi MS, Black BS, Celedon P, Leubitz A, Moghtaderi A, Nikolla DA, et al. Emergency department volume, severity, and crowding since the onset of the coronavirus disease 2019 pandemic. Ann Emerg Med. 2023;82(6):650-660.
doi pubmed - Moore R, Purvis RS, Hallgren E, Reece S, Padilla-Ramos A, Gurel-Headley M, Hall S, et al. "I am hesitant to visit the doctor unless absolutely necessary": A qualitative study of delayed care, avoidance of care, and telehealth experiences during the COVID-19 pandemic. Medicine (Baltimore). 2022;101(32):e29439.
doi pubmed - Czeisler ME, Marynak K, Clarke KEN, Salah Z, Shakya I, Thierry JM, Ali N, et al. Delay or avoidance of medical care because of COVID-19-Related concerns - United States, June 2020. MMWR Morb Mortal Wkly Rep. 2020;69(36):1250-1257.
doi pubmed - Findling MG, Blendon RJ, Benson JM. Delayed care with harmful health consequences-reported experiences from national surveys during coronavirus disease 2019. JAMA Health Forum. 2020;1(12):e201463.
doi pubmed - Rollins MK, Nelson AR, Boyd J, Couturier K, Joseph D, Burns K, Cone DC. Early changes in patient access interval during the COVID-19 pandemic. Prehosp Emerg Care. 2022;26(5):641-651.
doi pubmed - How COVID-19 has impacted our Nation’s EMS agencies. National Association of Emergency Medical Technicians. NAEMT.org. December 7, 2023. Available from: https://www.naemt.org/docs/default-source/covid-19/covid-impact-survey-06-03-2021.pdf.
- Wilcox ME, Rowan KM, Harrison DA, Doidge JC. Does unprecedented ICU capacity strain, as experienced during the COVID-19 pandemic, impact patient outcome? Crit Care Med. 2022;50(6):e548-e556.
doi pubmed - Mohr NM, Wessman BT, Bassin B, Elie-Turenne MC, Ellender T, Emlet LL, Ginsberg Z, et al. Boarding of critically ill patients in the emergency department. Crit Care Med. 2020;48(8):1180-1187.
doi pubmed - Kronibus N, Seiler F, Danziger G, Muellenbach RM, Reyher C, Becker AP, Kamphorst M, et al. Respiratory physiology of COVID-19 and influenza associated acute respiratory distress syndrome. J Clin Med. 2022;11(21):6237.
doi pubmed - Volkov L, Delpuech M, Conrad M, Courte G, Cravoisy A, Nace L, Baumann C, et al. Clinical outcomes and characteristics of critically ill patients with influenza- and COVID-19-induced ARDS: A retrospective, matched cohort study. Front Med (Lausanne). 2022;9:1027984.
doi pubmed - Rouze A, Martin-Loeches I, Povoa P, Makris D, Artigas A, Bouchereau M, Lambiotte F, et al. Relationship between SARS-CoV-2 infection and the incidence of ventilator-associated lower respiratory tract infections: a European multicenter cohort study. Intensive Care Med. 2021;47(2):188-198.
doi pubmed - Gjurasin B, Santini M, Krajinovic V, Papic N, Atelj A, Kotarski V, Krznaric J, et al. A retrospective comparison between influenza and COVID-19-associated ARDS in a Croatian tertiary care center. Wien Klin Wochenschr. 2021;133(7-8):406-411.
doi pubmed - Lee J, Lee YH, Chang HH, Choi SH, Seo H, Yoo SS, Lee SY, et al. Comparison of short-term mortality between mechanically ventilated patients with COVID-19 and influenza in a setting of sustainable healthcare system. J Infect. 2020;81(2):e76-e78.
doi pubmed - Leuchter RK, Delarmente BA, Vangala S, Tsugawa Y, Sarkisian CA. Health care staffing shortages and potential national hospital bed shortage. JAMA Netw Open. 2025;8(2):e2460645.
doi pubmed - Vaccination uptake, intent, and confidence. Centers for Disease Control and Prevention. Available from: https://www.cdc.gov/respvaxview/dashboards/vaccination-behavioral-social-drivers.html.
- Taylor CA, Whitaker M, Patton ME, Melgar M, Kirley PD, Kawasaki B, Yousey-Hindes K, et al. Trends in COVID-19-attributable hospitalizations among adults with laboratory-confirmed SARS-CoV-2-COVID-NET, June 2020 to September 2023. Influenza Other Respir Viruses. 2024;18(11):e70021.
doi pubmed - Brinkman S, de Keizer NF, de Lange DW, Dongelmans DA, Termorshuizen F, van Bussel BCT. Strain on scarce intensive care beds drives reduced patient volumes, patient selection, and worse outcome: a national cohort study. Crit Care Med. 2024;52(4):574-585.
doi pubmed - Neupane M, De Jonge N, Angelo S, Sarzynski S, Sun J, Rochwerg B, Hick J, et al. Measures and impact of caseload surge during the COVID-19 pandemic: a systematic review. Crit Care Med. 2024;52(7):1097-1112.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Clinical Medicine Research is published by Elmer Press Inc.