| Journal of Clinical Medicine Research, ISSN 1918-3003 print, 1918-3011 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Med Res and Elmer Press Inc |
| Journal website https://jocmr.elmerjournals.com |
Original Article
Volume 18, Number 4, April 2026, pages 271-281
Factors Predicting Diuretic Resistance in Patients With Acute Decompensated Heart Failure
Rarsari Soerarsoa, d , Dian Yaniarti Hasanaha
, Emir Yonasa
, Ahmad Pandu Pratamaa, Sunu Budhi Raharjoa
, Bambang Budi Siswantoa
, Maarten J.M. Cramerb
, Pim van der Harstb
, Marish I.F.J. Oerlemansc
aDepartment of Cardiology and Vascular Medicine, Faculty of Medicine Universitas Indonesia, National Cardiovascular Center Harapan Kita, Jakarta 11420, Indonesia
bDepartment of Cardiology, University Medical Center Utrecht, University of Utrecht, Utrecht 3584, the Netherlands
cDivision of Heart & Lungs, Department of Cardiology, University Medical Center Utrecht, University of Utrecht, Utrecht 3584, the Netherlands
dCorresponding Author: Rarsari Soerarso, Department of Cardiology and Vascular Medicine, Faculty of Medicine Universitas Indonesia, National Cardiovascular Center Harapan Kita, Jakarta 11420, Indonesia
Manuscript submitted September 18, 2025, accepted March 16, 2026, published online April 15, 2026
Short title: Factors Predicting Diuretic Resistance in ADHF
doi: https://doi.org/10.14740/jocmr6391
| Abstract | ▴Top |
Background: Acute decompensated heart failure (ADHF) is a leading cause of mortality and morbidity in the world. Diuretic resistance occurs in 20–30% of patients with ADHF and is an independent predictor of worsening clinical outcomes, immediate post-treatment death, and re-admission events. This study aims to: 1) identify factors that influence the occurrence of diuretic resistance in ADHF patients based on the underlying disease, comorbidities, vital signs, left ventricular ejection fraction, and laboratory parameters, and 2) investigate the clinical characteristics that serve as indicators of diuretic resistance incidence in patients with ADHF.
Methods: A retrospective cohort study was conducted on 535 patients treated with ADHF during the period from January to December 2019. Diuretic resistance was defined as a diuresis response of less than 1,400 mL in the first 24 h after administration of 40 mg of intravenous (IV) furosemide (or equivalent). Subjects were observed for 24 h post 40 mg IV furosemide for occurrence of diuretic resistance. Bivariate and multivariate analyses were performed to synthesize clinical scoring system to predict occurrence of diuretic resistance.
Results: Diuretic resistance occurs in 68% of patients. Independent predictors obtained from multivariate logistic regression analysis were: history of diabetes mellitus (DM, P = 0.013), history of using IV loop diuretics > 6 days (P = 0.002), oral loop diuretic dose > 80 mg/day (P = 0.006), left ventricular ejection fraction (LVEF) ≤ 49% (P = 0.002), blood urea nitrogen (BUN) ≥ 21 mg/dL (P < 0.001), and serum chloride < 98 mmol/L (P < 0.001). In addition, a scoring system has been made from the final model.
Conclusion: DM, history of IV loop diuretic, daily loop diuretic dosage, LVEF < 49%, BUN > 21 mg/dL, and serum chloride < 98 mmol/L were found to be statistically significant in association with occurrence of diuretic resistance using multivariate analysis and can be synthesized into a clinical scoring system to help predict diuretic resistance.
Keywords: Heart failure; Systolic heart failure; Cardiorenal syndrome; Heart decompensation; Congestive heart failure
| Introduction | ▴Top |
Heart failure (HF) remains a significant public health issue with high prevalence and incidence in both developing and developed countries. HF also contributes to a significant portion of admitted patients in these countries. Even with the advancement in the treatment of acute decompensated heart failure (ADHF), the rehospitalization and mortality rates remain high. It is estimated that 29–50% of patients with HF will experience rehospitalization in a year, with an intrahospital mortality rate of 3–4% [1, 2].
The population of ADHF patients in Indonesia possesses a rather different clinical presentation and characteristics compared to developed countries. Patients in Indonesia commonly present with a younger age, more severe clinical presentation, lower ejection fraction, and the common finding of diabetes mellitus (DM) as a comorbidity [1]. The primary manifestation of acute or chronic HF is still retention and accumulation of fluids that lead to congestion at the organ level. Loop diuretics have been proven to be effective in alleviating congestion in patients with ADHF, as has been recommended by various treatment guidelines [3–5].
The long-term use of loop diuretics might lead to diuretic resistance. Diuretic resistance is a phenomenon in which impaired sensitivity to diuretics occurs, leading to reduced diuresis and natriuresis response, which impedes euvolemia status in patients with chronic HF [3]. Studies have shown diuretic resistance to be 20–30% in patients with HF; diuretic resistance has also been shown to be predictive of rehospitalization and death [6–8]. However, there have not been any clinical indicators of diuretic resistance in HF patients. This study aims to investigate the clinical characteristics that serve as indicators of diuretic resistance incidence in patients with ADHF.
| Materials and Methods | ▴Top |
This is a retrospective cohort study that included all patients diagnosed with ADHF who were hospitalized in National Cardiovascular Center Harapan Kita (NCCHK), Jakarta, Indonesia, between January 1, 2019, and December 31, 2019 (Fig. 1). Data were gathered using medical records. The inclusion criteria were: 1) patients aged > 18 years or older; 2) hospitalized ADHF patients diagnosed with the presence of at least one clinical sign of ADHF (dyspnea, orthopnea, or edema), and one congestive symptom of ADHF (basilar rales, peripheral edema, ascites, increased jugular venous pressure), or any signs of pulmonary congestion from a chest X-ray. The exclusion criteria were: 1) ADHF secondary to congenital heart disease; 2) ADHF secondary to valvular structural abnormalities due to rheumatic heart disease; 3) ADHF with cardiogenic shock; 4) ADHF patients with chronic kidney disease (CKD) with estimated glomerular filtration rate (eGFR) < 15 mL/min/1.73 m2.
 Click for large image | Figure 1. Study flowchart for patients selection and analysis. |
Diuretic resistance was defined as a diuresis response of less than 1,400 mL after administration of 40 mg of intravenous (IV) furosemide equivalents in the first 24 h [7]. Diabetes is defined as a positive oral glucose tolerance test, HbA1c > 6.5%, or fasting plasma glucose > 126 mg/dL. CKD was defined as reductions in eGFR as per KDIGO recommendation, with GFR based on serum creatinine calculated using the CKD-EPI formula. Anamnesis, physical examinations, and laboratory panels were done in the emergency department at the patient’s initial presentation, while other data were retrospectively collected from medical records. Several numerical variables were transformed into categorical variables based on the current literature regarding diuretic resistance and ADHF. The sixth day of loop diuretic usage as an onset of diuretic resistance was chosen as threshold, based on several studies that observed hypertrophy and hyperplasia of renal distal convoluted tubule (DCT) and the occurrence of what is known as braking phenomenon that can be observed from the sixth day of loop diuretic administration [9–11]. Daily furosemide dose threshold of 80 mg is based on a study that defines diuretic resistance as persistent congestion necessitating hospitalization in patients already receiving daily doses of 80 mg of furosemide [12]. We elected to choose 49% as a threshold for ejection fraction in our study based on the guideline published by the European Society of Cardiology, which differentiates heart failure with reduced ejection fraction (HFREF) and heart failure with moderately reduced ejection fraction at 49% of ejection fraction [5]. Laboratory values were dichotomized based on established reference ranges.
This study was written as per the guidelines of Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis (TRIPOD) guideline [13].
Data analysis
Categorical data were presented in frequency and proportion. Numerical data were evaluated with a normality test. Data were analyzed with IBM SPSS Statistics for Windows (Version 25.0; IBM Corp., Armonk, NY). Numerical data with normal distribution were presented in mean ± SD, while abnormally distributed numerical data were presented with median (min–max). Bivariate analyses were done using the Chi-square test and Fisher’s exact test between predictor variables of diuretic resistance. Multivariate analyses were done in bivariate variables with P values < 0.25. We performed logistic regression with the backward selection method. Results of the multivariate analysis were further tested with the Hosmer-Lemeshow test for calibration and receiver operating characteristics (ROC) for discrimination test, and optimal sensitivity and specificity were determined using ROC results (Fig. 1). The final multivariate model was transformed into a scoring system using the B coefficient and standard error values. Variables that were included in the scoring system were weighed using the B coefficient and standard error values and were reapplied to the overall study subjects. The final scoring system model was then analyzed against the outcome of the study using logistic regression to determine the incidence probability of the study outcome using the proposed scoring system. The proposed scoring system was then analyzed using the area under the curve (AUC) method to determine sensitivity, specificity, and cutoff value using optimum specificity and sensitivity values.
Ethical clearance
Ethical clearance for this study was issued by the Institutional Ethical Review Board of NCCHK (Decision Letter Number LB.02.01/VII.450/KEP.055/2020).
| Results | ▴Top |
A total of 535 patients were included in this study between January and December 2019 at NCCHK. Data from 579 samples were initially gathered from the hospital medical record system; 44 samples were excluded due to incomplete data. Baseline (bivariate) characteristics analyses were performed on all subjects. Multivariate analyses were done in bivariate variables with P-values < 0.25. We performed logistic regression, which was then used to construct a predictive model.
The study subjects consisted of ADHF patients; DM was found in 79.4% of the study subjects. Non-dialysis-dependent CKD was found in 6.9% of subjects. Diuretic resistance was found in 68.2% of study subjects. Due to the abnormal distribution of data, several variables were expressed in medians. There were 79.1% male subjects in this study, and no statistically significant difference in diuretic resistance was observed across genders. We observed higher medians of age in patients with diuretic resistance, and more patients with DM, CKD, use of loop diuretics > 6 days, and daily dose of loop diuretics > 80 mg (11.8% vs. 0.6%, P < 0.001) were found to have diuretic resistance. Lower systolic and diastolic blood pressure was also observed in patients with diuretic resistance compared to those without. On echocardiographic parameters, we observed higher medians of left ventricular ejection fraction (LVEF) and tricuspud annular point of systolic excursion (TAPSE) in the group without diuretic resistance. Most of the laboratory parameters in this study were observed to have a significant difference between groups with diuretic resistance and without diuretic resistance, such as blood urea nitrogen (BUN), serum creatinine, eGFR, serum sodium, and serum chloride (Table 1).
 Click to view | Table 1. Baseline Characteristics of Study Subjects |
Continuous variables were dichotomized before bivariate analysis based on median values and established clinical reference ranges. We then performed bivariate analysis between variables and incidence of diuretic resistance in patients, using Chi-square methods, and we calculated the odds ratio (OR), 95% confidence interval (CI), and P-value. P value of 0.25 was set as the threshold for inclusion in multivariate analysis (performed on all subjects, n = 535). Based on our results, variables that satisfy this threshold were age > 60 years (P = 0.011), DM (P < 0.001), CKD (P = 0.043), history of IV loop diuretic use > 6 days (P < 0.001), daily loop diuretic dose > 80 mg (P < 0.001), LVEF < 49% (P < 0.001), serum creatinine > 1.5 mg/dL (P < 0.001), eGFR < 59 mL/min/1.73 m2 (P < 0.001), BUN > 21 mg/dL (P < 0.001), serum sodium < 135 mmol/L (P = 0.002), and serum chloride < 98 mmol/L (P < 0.001) (Table 2). There were 198 patients (37%) who received a sub-maintenance dose of IV loop diuretics within 6 h of initial presentation, and 75.8% of these patients were diuretic-resistant (OR 1.773, 95% CI 1.196–2.629; P = 0.004).
 Click to view | Table 2. Bivariate Analysis Result |
The final model of our multivariate analysis included DM, use of loop IV loop diuretics > 6 days, daily loop diuretic dose > 80 mg, LVEF < 49%, BUN > 21 mg/dL, and serum chloride < 98 mmol/L (Table 3). We performed the Hosmer-Lemeshow test on our scoring/prediction model, which generated a result of 0.823, a Chi-square value of 4.368 signifying a well-calibrated model that can be used to infer a causal relationship between independent and dependent variables, which we tested further using AUC model analysis, generating an AUC result of 0.767 (95% CI 0.725–0.809) (Fig. 2). In creating the scoring system, we performed weighing of variables in our final multivariate model using the B coefficient/standard error ratio (Table 4). These results were subsequently used to create a score weight for the presence of each corresponding variable in the final proposed scoring system (Table 5).
 Click to view | Table 3. Final Model of Multivariate Analysis |
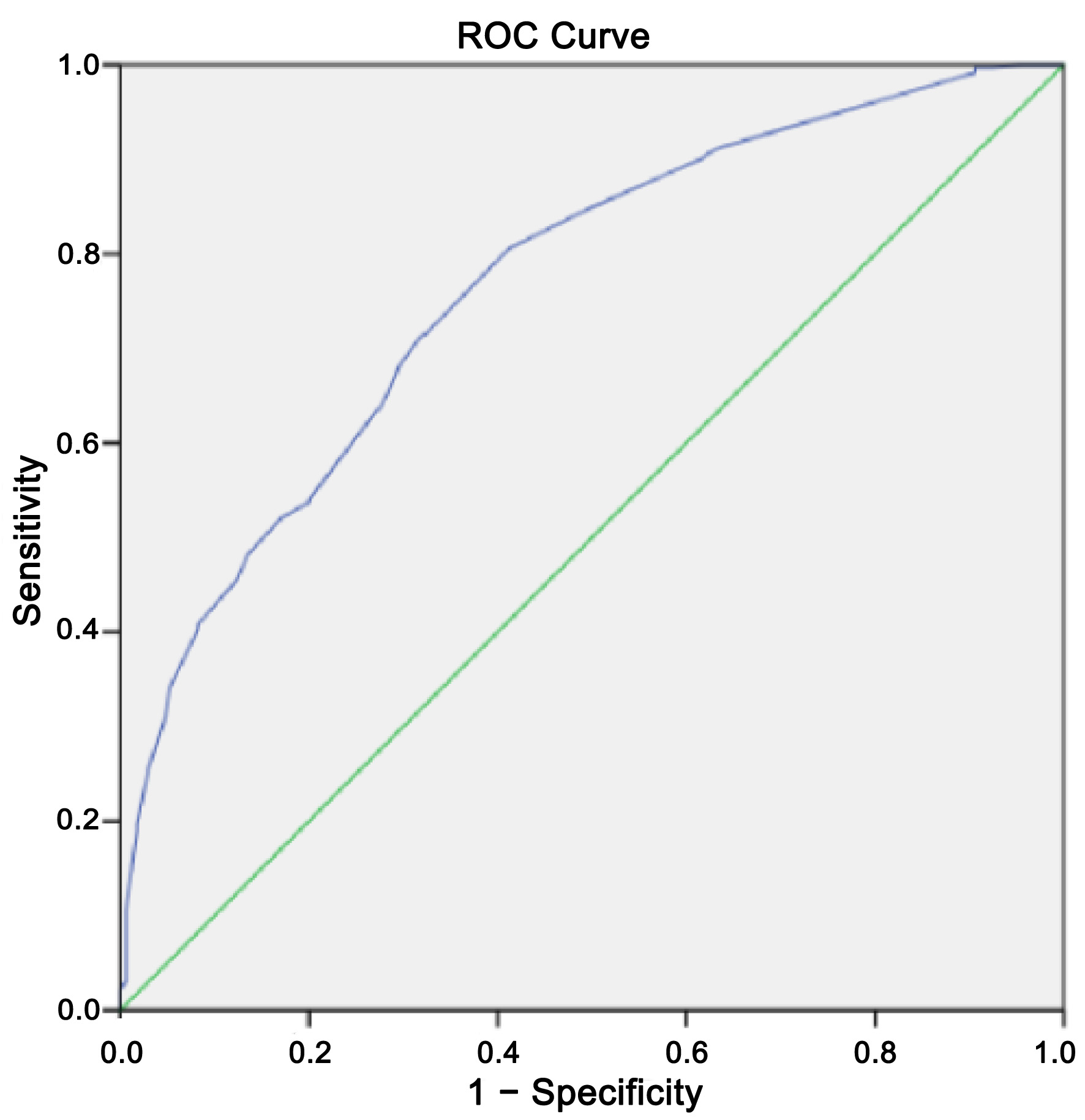 Click for large image | Figure 2. AUC result for the proposed diuretic resistance scoring system. AUC: area under the curve. |
 Click to view | Table 4. Scoring System Weighing |
 Click to view | Table 5. Final Proposed Score Model |
Our final predictive model consists of: 1) DM, 2) history of IV loop diuretic use > 6 days, 3) daily loop diuretic dosage > 80 mg, 4) LVEF ≤ 49%, and 5) BUN ≥ 21 mg/dL.
The probability of diuretic resistance based on our proposed scoring system was calculated with linear regression with a specificity and probability threshold of 95%, based on the presence of predictive variables on the patient being assessed. The results showed a low risk of diuretic resistance on a score of 1–3 (39–65% probability), moderate risk of diuretic resistance on score 4–6 (76–91% probability), and high risk of diuretic resistance on score 7–8 (95–96.4% probability). The sensitivity and specificity for our current predictive models were 59.7% and 72.4%, respectively (Table 6).
 Click to view | Table 6. Scoring System Interpretation |
| Discussion | ▴Top |
The main findings of this study are: 1) DM, history of IV loop diuretic, daily loop diuretic dosage, LVEF < 49%, BUN > 21 mg/dL, and serum chloride < 98 mmol/L were found to be statistically significant in association with occurrence of diuretic resistance using multivariate analysis. 2) Based on these findings, a scoring system with a fair performance (AUC 0.767 (0.725–0.809)) could be proposed to help quantitively predict the incidence of diuretic resistance in hospitalized ADHF patients.
In our study, diuretic resistance is defined as a urinary diuretic response of less than 1,400 mL per 40 mg of furosemide or equivalent. This definition was chosen based on a study on the ASCEND HF trial population by ter Maaten et al. The authors of this study concluded that an early assessment of diuretic response is as reliable as measurement after multiple days, thus providing clinical applicability in early identification of a diuretic-resistant patient [7, 14].
In this study, we observed a 68.2% prevalence of diuretic resistance out of 535 study subjects. This result is different from other studies regarding diuretic resistance in ADHF patients, which show a prevalence ranging from 20% to 35%. This finding might be caused by several factors such as the lower rates of loop diuretic administration in the first 6 h in this study and the different baseline characteristics of subjects in our study (198 patients (37%) received an initial sub-maintenance dose of IV loop diuretics within 6 h of initial presentation, 75.8% of these patients were diuretic-resistant (OR 1.773, 95% CI 1.196–2.629; P = 0.004)). This finding reflects a gap between guideline-directed medical therapy and real-world practices and challenges. We acknowledge that this inertia to fully implement guideline might mimic true diuretic resistance.
The higher rates of diuretic resistance in our study may also be caused by the advanced disease state of patients at presentation (median LVEF of 23% in diuretic resistance patients and median LVEF of 25% across all study subjects). Furthermore, higher diuretic resistance rates in our study subjects might also be caused by pseudo-resistance that is driven by initial suboptimal dosing. The European Society of Cardiology and the American Heart Association have recommended the administration of IV loop diuretics at 1–2.5 times the oral dose at the initial 6 h of presentation in patients with ADHF previously on oral loop diuretics [4, 15]. This recommendation stems from the decreased natriuretic and diuretic response to loop diuretics in a state of ADHF, which necessitates a higher loop diuretic dose. Inadequate IV dosing might even lead to decreased natriuretic and diuretic response, which will contribute to the incidence of diuretic resistance in ADHF patients [3].
Compared to previous studies on Western patients, we observed a lower median LVEF in our diuretic-resistant patients (23% (13–79%)). This finding is lower compared to patients’ LVEF in previous studies on ADHF diuretic resistance by Voors et al (37.9%), Valente et al (32.3%), and Trullas et al (50%). Compared to these studies, our subjects were also significantly younger with a median age of 58 years in the diuretic-resistant group, while the median age of patients was 72 years in the study by Voors et al, 70 years in the study by Valente et al, and 81 years in the study by Trullas et al [12, 16, 17]. The baseline characteristics of our study regarding age and LVEF were similar to a previous study by Siswanto et al which showed a trend of younger age (mean age of 60 years) and lower LVEF (mean LVEF of 33%) in Indonesian patients with ADHF, while patients in our country show lower trends of daily furosemide dose compared to Western countries [18]. While several other studies have studied the utilization of multiple agents to help curb diuretic resistance [14, 19], our study focused on synthesizing a new clinical scoring system using simple measurements to help better predict the occurrence of diuretic resistance in ADHF HFREF patients with type 2 DM. With early identification, diuretic-resistant patients might benefit from alternative treatment strategies to help overcome this resistance, ultimately leading to better outcomes for these patients.
More patients with diabetes developed diuretic resistance in our study. Diabetes has previously been known as a predictor of diuretic resistance [12, 17]. Diabetes causes increased RAAS activation, which necessitates the use of a higher loop diuretic dose to overcome this state. Insulin resistance in DM causes sodium retention in the kidneys through stimulation of Na+-K+-2CL- and Na-K-ATPase cotransporters.
The use of loop diuretics causes specific changes in the microstructure of the kidneys. Hypertrophy and hyperplasia of the distal tubules of the kidneys have been found to occur on the sixth to eighth day of initial loop diuretic exposure; this phenomenon is known as a braking phenomenon [10, 11, 20]. In this study, based on this assumption, we found supporting findings that a history of diuretic dosage > 6 days is a predictor of diuretic resistance in ADHF patients. Theoretically, the braking phenomenon secondary to loop diuretic use is one of the contributing factors to diuretic resistance, which causes a positive correlation between loop diuretic dose and diuretic resistance onset. Although a history of diuretic usage > 6 days was identified as a strong predictor in our study, the retrospective nature of this study makes it rather challenging to establish definitive causality. Despite the fact that these findings are in accordance with the physiological “braking phenomenon” secondary to distal tubule hypertrophy, we acknowledge the possibility of reverse causality in which poorer diuretic response inadvertently necessitates longer decongestion efforts using loop diuretics. However, it should be noted that, regardless of this, a prolonged diuretic requirement is very useful to identify high-risk patients who might need more intense diuretic decongestion therapy.
A study by Trullas et al defined diuretic resistance as persistent congestion that necessitates hospital admission even with oral loop diuretics > 80 mg/day [12]. Based on our analysis, we found that the use of oral loop diuretics > 80 mg/day was strongly predictive of diuretic resistance (OR 15.107, 95% CI 2.265–129.177). The presence of DM as a comorbidity also poses a significant risk for diuretic resistance. This is due to the risk of nephropathy of DM itself; furthermore, a recent study has found a relationship between diabetic kidney disease and worsening in HF. In their study, Sharma et al described that microvascular derangements in both kidney and heart microvasculature might be the cause of increased risk of congestion in diabetic HF patients [21].
The study by Trullas et al also showed lower LVEF in patients with diuretic resistance [12]. The decreased cardiac output secondary to reduced LVEF activates RAAS, AVP, and the autonomic nervous system, which in turn causes retention of fluid and sodium, which ultimately dampens the kidney’s response to loop diuretics. In this study, we also found LVEF < 49% to be predictive of diuretic resistance (OR 3.186, 95% CI 1.530–6.633; P = 0.002).
BUN has been previously found to be a predictor of diuretic resistance [16, 17]. BUN has competitive properties against loop diuretics, especially furosemide, at the organic anion transporters (OAT) of the kidneys. The increase in BUN levels will cause reductions of binding between serum loop diuretics and OAT, which will contribute to the incidence of diuretic resistance. In this study, BUN > 21 mg/dL is found to be predictive of diuretic resistance (OR 2.477, 95% CI 1.641–3.740; P < 0.001). Hypochloremia is also found to be predictive of diuretic resistance in this study (OR 2.699, 95% CI 1.672–4.357; P < 0.001). Hypochloremia causes the release of renin and RAAS activation, which causes phosphorylation of with-no-lysine (WNK) kinase, which in turn will cause activation of Na+-K+-CL− cotransporter that will reabsorb sodium and dampens effectivity of loop diuretics [22–24].
Although comorbidities commonly increase and worsen as age advances, we did not find age to be a predictor of diuretic resistance. Our age cutoff of 58 years was based on a previous study by Siswanto et al on the characteristics of Indonesian patients with ADHF. This caused a lower threshold of age compared to previous studies on ADHF [12, 16, 17].
We included TAPSE in our multivariate model to better reflect any alterations of right heart function in our study subjects, considering that right heart dysfunction was prevalent in patients with HFREF [25–27]. In this study, we observed 464 subjects (86.7%) with LVEF < 30% and 156 subjects (29%) with LVEF < 30% and criteria of advanced HF. However, TAPSE was not found to be statistically significant during our multivariate analysis (P = 0.787).
We did not find eGFR and serum creatinine to be statistically significant predictors of diuretic resistance in our multivariate analysis. This is similar to previous studies dealing with diuretic resistance and cardiorenal syndrome, which describe renal dysfunction in the spectrum of HF as a secondary condition to the worsening of heart function. Reduced eGFR and elevated serum creatinine as markers of kidney dysfunction in cardiorenal syndrome signify reduced renal perfusion, not a primary worsening of the kidneys. In patients with concurrent CKD and HF, the fall in eGFR was thought to result from a decline in cardiac output; an even further reduction in renal function might be seen in states of reduced renal perfusion, such as in ADHF before treatment [28, 29]. We did not find hyponatremia to be associated with diuretic resistance; this is also the finding of several previous studies [22–24]. In contrast with a previous study [16], we did not find hypokalemia to be predictive of diuretic resistance in our study.
There were more diuretic resistance incidences observed in patients with hypochloremia in this study (83.6% vs. 16.4%, P < 0.001). Our findings are similar to other studies in which hypochloremia is associated with diuretic resistance and increased mortality in ADHF patients [22, 30–33].
In ADHF patients, hypochloremia is commonly seen and associated with lower diuretic response and less decongestion. Decreased levels of serum chloride sensed by macula densa lead to reabsorption of electrolytes and fluid reabsorption through upregulation of NKCC and NCC receptors. With these receptors being the same receptors which loop diuretics act on, upregulation of these receptors leads to a blunted response to loop diuretics in HF patients [34]. Activation of this sodium retentive system occurs as a counter-regulatory response in ADHF patients treated with loop diuretics and causes significant changes in the pharmacokinetics of loop diuretics in ADHF [35]. This correlates with our multivariate model findings in which hypochloremia is associated with diuretic resistance (OR 2.699, 95% CI 1.672–4.357; P < 0.001).
With an AUC of 0.767, we expect our proposed scoring system to perform fairly in predicting the incidence of diuretic resistance. With this AUC value, this scoring system posed an acceptable discriminatory capability to predict the incidence of diuretic resistance at admission [36, 37]. However, we were unable to do a comparison since no other diuretic resistance predictive score has been proposed. A score of 5 in our scoring system was shown to be the point at which sensitivity and specificity balance (59.7% sensitivity and 72.4% specificity). We chose a probability threshold of 95% in determining high-risk criteria of diuretic resistance in our scoring system, which correlates to a score of 7–8 on the score (95% specificity). This scoring system is intended to help differentiate between diuretic responders and nonresponders at admission. In peripheral hospitals, this scoring system can help in early recognition of the need for referral in cases with anticipated diuretic resistance.
Conclusion
DM, history of IV loop diuretic, daily loop diuretic dosage, LVEF < 49%, BUN > 21 mg/dL, and serum chloride < 98 mmol/L were found to be statistically significant in association with the occurrence of diuretic resistance using multivariate analysis. Based on these findings, a new scoring system with a fair performance could predict diuretic resistance among patients hospitalized with ADHF.
Study limitations
This study has several limitations. First, our dataset does not include detailed information regarding previous routine medications of ADHF subjects, such as angiotensin receptor blockers, angiotensin-converting enzyme inhibitors, beta blockers, and mineralocorticoid receptor antagonists, which might influence diuretic responsiveness. Furthermore, our retrospective study utilized data from January to December 2019, which precedes the widespread recommendation and use of sodium-glucose cotransporter-2 (SGLT2) inhibitors for HF treatment. Consequently, SGLT2 inhibitor usage was negligible in our cohort and was not included as a variable. The presence of DM in this study was defined binarily; data regarding the profiling of long-term diabetic control and microvascular complications were not recorded during emergency admission. Specific laboratory parameters that dynamically reflect intravascular volume status, such as serial hematocrit or serum osmotic pressure, were not uniformly available in this retrospective dataset, limiting our ability to quantify baseline hydration status. As a retrospective observational study, our data reflect everyday clinical realities rather than the strictly controlled environment of trial conditions, and initial loop diuretic dosing was at the discretion of the treating physician; consequently we acknowledge that the clinical inertia regarding optimal initial loop diuretic dosing might mimic true physiological resistance; however, this inertia provides valuable insight to the challenges of an ideal implementation of HF guidelines. Finally, although our proposed scoring system demonstrates good calibration and fair discriminative capability (AUROC 0.767 and Hosmer-Lemeshow test), a further prospective validation in larger multicenter cohorts will be needed to further explore the clinical utility and generalizability of this scoring system.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
The authors declare that they have no financial conflict of interest with regard to the content of this manuscript.
Informed Consent
Informed consents were obtained from all study participants.
Author Contributions
Rarsari Soerarso: conceptualization, project administration, writing – original draft. Emir Yonas: formal analysis, writing – original draft. Ahmad Pandu Pratama: formal analysis, writing – original draft. Dian Yaniarti Hasanah: writing – review & editing. Sunu Budhi Raharjo: writing – review & editing. Bambang Budi Siswanto: writing – review & editing. Marish I.F.J Oerlemans: writing – review & editing. Pim van der Harst: writing – review & editing. Maarten J.M. Cramer: writing – review & editing.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
ADHF: acute decompensated heart failure; AUC: area under the curve; BUN: blood urea nitrogen; CAD: coronary artery disease; CKD: chronic kidney disease; DBP: diastolic blood pressure; DM: diabetes mellitus; eGFR: estimated glomerular filtration rate; LVEF: left ventricular ejection fraction; ROC: receiver operating curve; TAPSE: tricuspud annular point of systolic excursion
| References | ▴Top |
- Siswanto BB, Radi B, Radi B, et al. Heart Failure in NCVC Jakarta and 5 hospitals in Indonesia. Glob Heart. 2010;5(1):35.
doi - Lombardi C, Peveri G, Cani D, Latta F, Bonelli A, Tomasoni D, Sbolli M, et al. In-hospital and long-term mortality for acute heart failure: analysis at the time of admission to the emergency department. ESC Heart Fail. 2020;7(5):2650-2661.
doi pubmed - Ellison DH, Felker GM. Diuretic treatment in heart failure. N Engl J Med. 2017;377(20):1964-1975.
doi pubmed - Mullens W, Damman K, Harjola VP, Mebazaa A, Brunner-La Rocca HP, Martens P, Testani JM, et al. The use of diuretics in heart failure with congestion - a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2019;21(2):137-155.
doi pubmed - Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS, Falk V, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC)Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2016;37(27):2129-2200.
doi pubmed - Jardim SI, Ramos Dos Santos L, Araujo I, Marques F, Branco P, Gaspar A, Fonseca C. A 2018 overview of diuretic resistance in heart failure. Rev Port Cardiol (Engl Ed). 2018;37(11):935-945.
doi pubmed - ter Maaten JM, Dunning AM, Valente MA, Damman K, Ezekowitz JA, Califf RM, Starling RC, et al. Diuretic response in acute heart failure-an analysis from ASCEND-HF. Am Heart J. 2015;170(2):313-321.
doi pubmed - Shah N, Madanieh R, Alkan M, Dogar MU, Kosmas CE, Vittorio TJ. A perspective on diuretic resistance in chronic congestive heart failure. Ther Adv Cardiovasc Dis. 2017;11(10):271-278.
doi pubmed - De Bruyne LK. Mechanisms and management of diuretic resistance in congestive heart failure. Postgrad Med J. 2003;79(931):268-271.
doi pubmed - Kaissling B, Stanton BA. Adaptation of distal tubule and collecting duct to increased sodium delivery. I. Ultrastructure. Am J Physiol. 1988;255(6 Pt 2):F1256-1268.
doi pubmed - Loon NR, Wilcox CS, Unwin RJ. Mechanism of impaired natriuretic response to furosemide during prolonged therapy. Kidney Int. 1989;36(4):682-689.
doi pubmed - Trullas JC, Casado J, Morales-Rull JL, Formiga F, Conde-Martel A, Quiros R, Epelde F, et al. Prevalence and outcome of diuretic resistance in heart failure. Intern Emerg Med. 2019;14(4):529-537.
doi pubmed - Collins GS, Reitsma JB, Altman DG, Moons KG. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement. BMJ. 2015;350:g7594.
doi pubmed - Dandamudi S, Chen HH. The ASCEND-HF trial: an acute study of clinical effectiveness of nesiritide and decompensated heart failure. Expert Rev Cardiovasc Ther. 2012;10(5):557-563.
doi pubmed - Felker GM, Ellison DH, Mullens W, Cox ZL, Testani JM. Diuretic therapy for patients with heart failure: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75(10):1178-1195.
doi pubmed - Voors AA, Davison BA, Teerlink JR, Felker GM, Cotter G, Filippatos G, Greenberg BH, et al. Diuretic response in patients with acute decompensated heart failure: characteristics and clinical outcome—an analysis from RELAX-AHF. Eur J Heart Fail. 2014;16(11):1230-1240.
doi pubmed - Valente MA, Voors AA, Damman K, Van Veldhuisen DJ, Massie BM, O'Connor CM, Metra M, et al. Diuretic response in acute heart failure: clinical characteristics and prognostic significance. Eur Heart J. 2014;35(19):1284-1293.
doi pubmed - Siswanto BB, Radi B, Kalim H, et al. Heart Failure in NCVC Jakarta and 5 hospitals in Indonesia. CVD Prev Control. 2010;5(1):35-38.
doi - Bohn BC, Hadgu RM, Pope HE, Shuster JE. Oral metolazone versus intravenous chlorothiazide as an adjunct to loop diuretics for diuresis in acute decompensated heart failure with reduced ejection fraction. Hosp Pharm. 2019;54(6):351-357.
doi pubmed - Nag DC, Murshed A, Chakrabortty R, Rahman MR. Mechanism and management of diuretic resistance in congestive heart failure. Journal of Dhaka National Medical College & Hospital. 2012;17(1):44-46.
doi - Sharma A, Inzucchi SE, Testani JM, Ofstad AP, Fitchett D, Mattheus M, Verma S, et al. Kidney and heart failure events are bidirectionally associated in patients with type 2 diabetes and cardiovascular disease. ESC Heart Fail. 2024;11(2):737-747.
doi pubmed - Ter Maaten JM, Damman K, Hanberg JS, Givertz MM, Metra M, O'Connor CM, Teerlink JR, et al. Hypochloremia, diuretic resistance, and outcome in patients with acute heart failure. Circ Heart Fail. 2016;9(8).
doi pubmed - Testani JM, Hanberg JS, Arroyo JP, Brisco MA, Ter Maaten JM, Wilson FP, Bellumkonda L, et al. Hypochloraemia is strongly and independently associated with mortality in patients with chronic heart failure. Eur J Heart Fail. 2016;18(6):660-668.
doi pubmed - Hanberg JS, Rao V, Ter Maaten JM, Laur O, Brisco MA, Perry Wilson F, Grodin JL, et al. Hypochloremia and diuretic resistance in heart failure: mechanistic insights. Circ Heart Fail. 2016;9(8).
doi pubmed - Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, Flachskampf FA, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2015;16(3):233-270.
doi pubmed - Iglesias-Garriz I, Olalla-Gomez C, Garrote C, Lopez-Benito M, Martin J, Alonso D, Rodriguez MA. Contribution of right ventricular dysfunction to heart failure mortality: a meta-analysis. Rev Cardiovasc Med. 2012;13(2-3):e62-69.
doi pubmed - Crespo-Leiro MG, Metra M, Lund LH, Milicic D, Costanzo MR, Filippatos G, Gustafsson F, et al. Advanced heart failure: a position statement of the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2018;20(11):1505-1535.
doi pubmed - Weinfeld MS, Chertow GM, Stevenson LW. Aggravated renal dysfunction during intensive therapy for advanced chronic heart failure. Am Heart J. 1999;138(2 Pt 1):285-290.
doi pubmed - Ljungman S, Laragh JH, Cody RJ. Role of the kidney in congestive heart failure. Relationship of cardiac index to kidney function. Drugs. 1990;39(Suppl 4):10-21; discussion 22-14.
doi pubmed - Grodin JL, Simon J, Hachamovitch R, Wu Y, Jackson G, Halkar M, Starling RC, et al. Prognostic role of serum chloride levels in acute decompensated heart failure. J Am Coll Cardiol. 2015;66(6):659-666.
doi pubmed - Grodin JL, Sun JL, Anstrom KJ, Chen HH, Starling RC, Testani JM, Tang WH. Implications of serum chloride homeostasis in acute heart failure (from ROSE-AHF). Am J Cardiol. 2017;119(1):78-83.
doi pubmed - Marchenko R, Sigal A, Wasser TE, Reyer J, Green J, Mercogliano C, Khan MS, et al. Hypochloraemia and 30 day readmission rate in patients with acute decompensated heart failure. ESC Heart Fail. 2020;7(3):903-907.
doi pubmed - Kondo T, Yamada T, Tamaki S, Morita T, Furukawa Y, Iwasaki Y, Kawasaki M, et al. Serial change in serum chloride during hospitalization could predict heart failure death in acute decompensated heart failure patients. Circ J. 2018;82(4):1041-1050.
doi pubmed - Rivera FB, Alfonso P, Golbin JM, Lo K, Lerma E, Volgman AS, Kazory A. The role of serum chloride in acute and chronic heart failure: a narrative review. Cardiorenal Med. 2021;11(2):87-98.
doi pubmed - Masella C, Viggiano D, Molfino I, Zacchia M, Capolongo G, Anastasio P, Simeoni M. Diuretic resistance in cardio-nephrology: role of pharmacokinetics, hypochloremia, and kidney remodeling. Kidney Blood Press Res. 2019;44(5):915-927.
doi pubmed - Mandrekar JN. Receiver operating characteristic curve in diagnostic test assessment. J Thorac Oncol. 2010;5(9):1315-1316.
doi pubmed - Janssens A, Martens FK. Reflection on modern methods: Revisiting the area under the ROC Curve. Int J Epidemiol. 2020;49(4):1397-1403.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Clinical Medicine Research is published by Elmer Press Inc.