| Journal of Clinical Medicine Research, ISSN 1918-3003 print, 1918-3011 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Med Res and Elmer Press Inc |
| Journal website https://jocmr.elmerjournals.com |
Review
Volume 18, Number 3, March 2026, pages 142-156
The Correct Nutritional Intake in the Prevention and Treatment of Skin Lesions in Patients With Spinal Cord Injury
Alessandra Arenia, h, William Capecib, Alessandro Corsic, Giulio Del Popolod, Luisa De Palmae, Laura Pelizzarif, Primo Vercillig
aAUSL Bologna, Bologna, Italy
bSpinal Cord Unit, Department of Neurogical Science, University Hospital of Marche, Ancona, Italy
cB. Braun SpA, Milan, Italy
dSpinal Unit, Department of Neuroscience and Translational Medicine, Azienda Ospedaliero-Universitaria Policlinico, Bari, Italy
eDepartment of Neuro-Urology & Spinal Unit, Azienda Ospedaliera-Universitaria Careggi, Firenze, Italy
fSpinal Unit and Neurorehabilitation, Department of Rehabilitation Medicine, AUSL Piacenza, Piacenza, Italy
gAmbulatorio di Nutrizione, Poliambulatorio Chiros, Forli, Italy
hCorresponding Author: Alessandra Areni, AUSL Bologna, Bologna, Italy
Manuscript submitted October 3, 2025, accepted January 10, 2026, published online March 26, 2026
Short title: Nutritional Prevention of Skin Lesions in SCI
doi: https://doi.org/10.14740/jocmr6399
| Abstract | ▴Top |
Background: Pressure injuries (PIs) are a serious and highly prevalent complication in individuals with spinal cord injury (SCI), negatively impacting quality of life, rehabilitation, and healthcare costs. Given their multifactorial etiology, a multidisciplinary approach that includes nutritional interventions is essential. This review provides expert consensus on the role of nutrition in PI prevention and management in SCI patients.
Methods: A panel of clinicians, including experts in wound care, nutrition, urology, internal medicine, and rehabilitation, assessed current practices and proposed evidence-based dietary recommendations. A narrative literature review supported the consensus process.
Results: PI risk screening through SCI-specific tools (e.g., Braden scale) and shared decision-making were supported. Nutritional recommendations include aligning energy intake to reduced total daily energy expenditure, highly digestable protein rich in essential amino acids, omega-3 fatty acids, and micronutrients, with adequate hydration. Dietary patterns should be anti-inflammatory, rich in fiber, fruits/vegetables, legumes, fish, and fermented foods. Also, they should modulate gut microbiota and reduce advanced glycation end products through food choices and cooking methods. Practical tools, the Food Suitability Map (traffic-light guidance) and food diary, were proposed to support education, adherence, and self-management. Implementation guidance is provided for acute, chronic, community, and readmission/surgical settings.
Conclusions: This review stresses the urgent need for standardized, SCI-specific nutritional protocols and enhanced interdisciplinary collaboration. Nutrition should be integrated into routine PI care to improve patient outcomes and reduce healthcare burden. Future research should explore nutraceuticals, refine existing protocols, assess long-term impacts of dietary strategies, and strengthen implementation in clinical practice.
Keywords: Spinal cord injury; Pressure injuries; Nutrition therapy; Wound healing; Gut microbiota; Inflammation; Nutrient supplementation
| Introduction | ▴Top |
Spinal cord injury (SCI) is defined as any damage to the spinal cord resulting in partial or complete loss of motor, sensory, and autonomic function below the level of injury. Higher-level injuries, particularly cervical SCIs, lead to tetraplegia and profound immobility, severely limiting functional independence. Thoracic or lumbar SCIs typically result in paraplegia, with preserved upper limb function but substantial limitations in lower-body mobility [1]. SCI leads to complications that significantly impact quality of life (QoL), including respiratory dysfunction, neurogenic bowel and bladder, osteoporosis, pressure injuries (PIs), and alterations in metabolism that lead to changes in body composition such as sarcopenia and obesity [2–6]. Tetraplegia profoundly increases the risk of malnutrition due to reduced muscle mass, impaired gastrointestinal function, and greater metabolic alterations and paraplegia contributes to reduced physical activity and altered nutrient requirements [7]. Neurogenic obesity occurs due to low metabolic rate and total daily energy expenditure (TDEE) [8] and increases cardiometabolic risk [9]. Also, gut microbiome changes that occur in SCI contribute to chronic inflammation and metabolic disorders [10]. Early after SCI, weight loss may occur due to increased metabolic demand from trauma [11] or undernutrition driven by infections, dysphagia, hormonal changes, wounds, and psychological stress [12].
PIs are localized skin or tissue injury from pressure or shear [13] and, despite being preventable, 20–50% of acute SCI patients develop PIs in the hospital [14]. PI annual incidence in SCI patients ranges from 25.3% to 41.0% [15] and lifetime risk reaches 85.0–95.0% [16]. Malnutrition, which affects 86% of SCI patients with PIs [17, 18], is a major risk factor for PI severity and delayed healing [19]. PIs cause infections, impair rehab, reduce well-being, extend hospital stays, and raise mortality [20–22]. They impose high healthcare costs ($4,745/month) [23], along with indirect costs and caregiver burden [24]. PIs may represent 25% of SCI care costs, with treatment more expensive than prevention [25, 26].
This review explores PI prevention and treatment strategies across care settings, emphasizing the role of targeted nutritional interventions in SCI.
| Methods | ▴Top |
A multidisciplinary panel, including a wound care physician, urologist, physician nutrition specialist, two internal medicine specialists, and two physical medicine and rehabilitation physicians, met in July 2024 to address PI management in SCI patients in Italy. The group evaluated PI risk and reviewed prevention and treatment strategies, with a focus on identifying nutritional deficiencies and proposing dietary interventions to support wound healing. This review presents consensus-based recommendations (≥ 70% agreement). A narrative literature review was also conducted to support the discussions. Articles were selected based on their relevance following targeted searches in PubMed. The search strategy included combinations of the following keywords and Boolean operators: “spinal cord injury” AND “pressure injury” OR “pressure ulcer” OR “pressure sores”; “spinal cord injury” AND “risk factors” OR “mobility limitations” OR “skin integrity” OR “autonomic dysreflexia”; “spinal cord injury” AND “risk assessment” OR “risk assessment tools” OR “Braden Scale” OR “SCIPUS” OR “SCIPUS-A” OR “LPD Scale”; “spinal cord injuries” AND “nutrition assessment” OR “nutritional status”; “spinal cord injury” AND “change in body composition” OR “malnutrition” OR “undernutrition” OR “dietary supplements”; “spinal cord injury” AND “energy expenditure”; “anti-inflammatory diets”; “anti-inflammatory nutrients”; “Mediterranean diet” AND “inflammation”; “spinal cord injury” AND “gastrointestinal microbiome” OR dysbiosis; “spinal cord injury” AND “multidisciplinary”.
| Results | ▴Top |
PI preventive strategies: a multi-faceted approach
Effective management of SCI patients should prioritize early PI risk assessment, the timely implementation of preventive strategies (e.g., specialized pressure redistribution devices) and early detection [25]. Preventing PIs requires a lifelong commitment, emphasizing the need for continuous education for patients and structured training programs for healthcare professionals (HCPs) [26–28]. As growing evidence underscores the critical role of nutrition in wound healing, targeted dietary interventions might support the prevention of PIs and tissue repair while reducing complications [29].
Comprehensive risk assessment, predictive tools for PIs, and PI management
Previously published data have categorized PI risk factors into intrinsic and extrinsic [30–32]. Building on this evidence and their clinical experience, the authors have drawn attention to the most relevant elements that increase vulnerability to PI development and delay wound healing (Table 1).
 Click to view | Table 1. Risk Factors for PIs in SCI Patients |
The authors have also emphasized the need for a collaborative approach between patients and the multidisciplinary team, including physical medicine and rehabilitation physicians, wound-care specialists, neurologists, internal medicine specialists, urologists, physician nutrition specialists, and nurses [33]. This model aims at improving prevention strategies and outcomes through shared decision-making. Also, access to relevant health information enhances adherence to preventive measures and supports effective self-care. Given patient variability, especially in acute phases, regular reassessment is needed. In community settings, where limited time and resources constrain HCPs to episodic contact, patients and their families are increasingly expected to manage their own care [34].
The authors reviewed the reliability of tools used to predict PI risk in SCI patients (Table 2 [31, 33, 35–39]).
 Click to view | Table 2. Existing Evidence on the Use of Risk Assessment Tools for PI Prediction in SCI Populations |
Four key themes have been identified regarding patient perceptions of PI risk and the communication of risk between nurse and patient in the home setting: 1) PI awareness; 2) importance of repositioning; 3) healthy eating; and 4) risk interpretation [40]. Accordingly, the authors stressed the need to educate HCPs in PI prevention, detection, and management, possibly through master’s degree programs in SCI-related PI care.
The authors underlined the importance of an assessment by wound-care expert physicians and/or nurses trained in wound care to classify the PI based on staging (Fig. 1) [41].
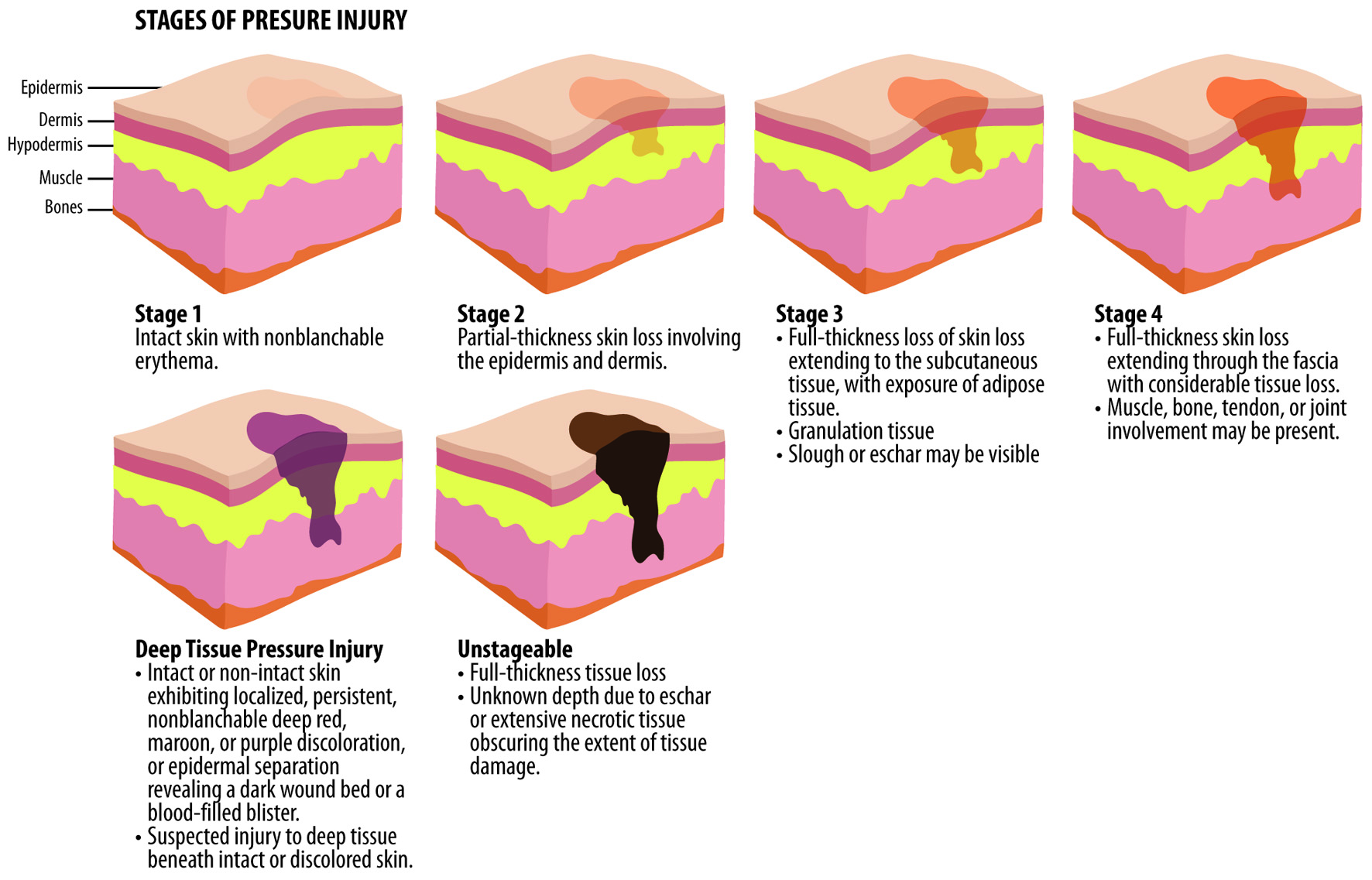 Click for large image | Figure 1. Stages of PIs based on EPUAP classification. EPUAP: European Pressure Ulcer Advisory Panel; PIs: pressure injuries. |
The importance of multidisciplinary pre- and postoperative care and patient education has been highlighted in prior research. This strategy is based on the collaboration between plastic surgeons, internal medicine specialists, and physical medicine and rehabilitation physicians [30]. Key clinical domains involved in the holistic management of patients with PIs, emphasizing a multidisciplinary approach, are outlined in Table 3 [30, 42–51].
 Click to view | Table 3. Comprehensive Multidisciplinary Assessment Domains for Patients With Chronic PIs |
The role of nutrition in wound healing of PIs in SCI patients
Although precise nutrient needs for SCI patients remain unclear, high-protein oral supplements have been shown to reduce PI incidence by 25% in at-risk individuals. Energy, protein, arginine, glutamine, omega-3 fatty acids, and key micronutrients, such as vitamins A, B, C, zinc, and iron, support collagen synthesis, inflammation control, and tissue repair [52, 53]. Essential amino acids aid in nitrogen balance and promote fibroblast proliferation, angiogenesis, and immune function. Adequate hydration maintains skin integrity, cellular metabolism and blood flow to injured tissue, preventing further skin damage and delayed wound healing [52, 53]. Malnutrition compromises collagen formation and immune response, worsening PI outcomes [54, 55]. Chronic wounds, such as PIs, sustain prolonged inflammation, with low growth factor levels and microbial contamination. Hypermetabolism and a catabolic state further raise protein needs and TDEE [56]. In this context, the intake of protein sources highly digestable and containing essential aminoacids is crucial [57, 58].
The nutritional needs of SCI patients and SCI patients with PIs
Published data reports that TDEE ranges from 2,030 to 3,344 kcal/day in the acute SCI phase, and from 1,332 to 2,834 kcal/day in chronic SCI. TDEE is reduced by up to 54% in individuals with tetraplegia and around 20% in those with paraplegia [9, 59]. Despite generally high protein intake, SCI patients may lack essential amino acids, which are vital for protein synthesis and tissue repair, particularly important in the context of PIs [60, 61]. Moreover, PI development rapidly depletes protein reserves, mainly skeletal muscle, accelerating malnutrition [62]. Accordingly, to identify patients at nutritional risk, the Spinal Nutrition Screening Tool (SNST), a validated, SCI-specific screening instrument, assesses skin integrity alongside several SCI-related and general clinical factors. These include age, weight history, appetite, injury level, comorbidities (e.g., need for artificial ventilation or nutrition), use of supplements, modified-texture diets, and the individual’s ability to eat. Each item is scored from 0 to 5, and the total score indicates the malnutrition risk category: 0–10 = low risk, 11–15 = moderate risk, and > 15 = high risk [63].
Guidelines recommend in SCI patients without PIs or infection an intake of 0.8–1.0 g/kg/day of protein to maintain protein status [62] and limiting saturated fat to 5–6% of total energy intake [64]. Carbohydrates should constitute 45–65% of daily energy intake, preferably from complex, fiber-rich, low-glycemic sources. A fiber intake of 25–30 g/day is advised to support metabolism and bowel function, and prevent neurogenic bowel dysfunction [62]. Although guidelines exist [9, 64, 65], there is an urgent need for innovative nutrition protocols tailored to SCI patients at risk of or presenting with PIs [66].
Emerging perspectives on diet in SCI
Tailored nutrition is essential for preventing PIs and promoting wound healing and should reflect specific clinical needs, such as metabolic syndrome or post-surgical recovery. Annual assessments and education by registered dietitians should be routine. Supplements may support outcomes but must follow individualized plans based on comprehensive evaluations addressing inflammation, oxidative stress, and immunity.
Diets should balance macronutrients to meet reduced TDEE and prioritize nutrient-dense, low-energy foods [67]. Given SCI-related immune dysfunction and inflammation, and comorbidities (e.g., diabetes and hypertension) [68], nutrition should aim to reduce advanced glycation end products (AGEs), which promote inflammation [69]. Although not SCI-specific, AGEs have been linked to degenerative cervical myelopathy [70]. Foods rich in melatonin or L-tryptophan (e.g., potatoes and chickpeas) possess anti-inflammatory properties and should be included [71, 72]. L-tryptophan aids serotonin and melatonin production and generates anti-inflammatory metabolites, such as indole-3-propionic acid via gut microbiota metabolism [73]. Similarly, L-arginine supports endothelial function, cardiovascular health, wound healing, and reduces inflammation [74].
Emerging perspectives on gut microbiome modulation in SCI
Recent studies link intestinal dysbiosis, in particular bacterial lipopolysaccharides (LPS), to SCI-related gastrointestinal dysfunctions, systemic low-grade inflammation, obesity, insulin resistance, diabetes, and cardiovascular disease [75, 76]. In SCI, LPS translocation worsens inflammation [77, 78], making intestinal barrier integrity crucial for preventing immune dysregulation and systemic complications [79]. Indeed, gut health and immunity are closely interconnected, and a balanced gut microbiome supports the production of short-chain fatty acids (SCFAs), which exert anti-inflammatory effects and regulate insulin [80]. Reduced microbial diversity, common in SCI, increases susceptibility to infections, such as Clostridium difficile [81] and multidrug-resistant Staphylococcus aureus (MRSA) [82]. Enhancing microbial diversity and SCFA levels involves greater intake of fruits, vegetables, fish, and dietary fibers, such as pectins, inulin and resistant starches [77, 83, 84]. Melatonin has also been shown to correct SCI-induced microbiota imbalances [85]. Specific strains, such as Lactobacillus and Bifidobacterium produce neuroactive compounds, such as gamma-aminobutyric acid (GABA). Those molecules influence both central and peripheral nervous systems and potentially reduce anxiety and depression symptoms [86].
Anti-inflammatory dietary components for enhancing immune function and gut health
The authors emphasized the importance of dietary modifications to harness the nutraceutical properties of food. Anti-inflammatory diets prioritize foods known to reduce chronic low-grade inflammation, a key driver of many non-communicable diseases. These dietary patterns emphasize fruits, vegetables, whole grains, legumes, nuts, seeds, olive oil, herbs and spices, and sources of omega-3 fatty acids, such as fish. Conversely, they limit red and processed meat, refined carbohydrates, added sugars and saturated fats, which are linked to chronic low-grade inflammation [87]. Such diets are also rich in polyphenols, antioxidants, vitamins, minerals, fiber, and healthy fats, all of which modulate inflammatory pathways, by lowering pro-inflammatory cytokines and oxidative stress. Evidence suggests that anti-inflammatory diets, including the Mediterranean diet, can help reduce inflammation-related risk across a range of conditions, from obesity to cancer and cognitive decline, by improving metabolic health, supporting gut microbiota, and dampening the molecular signals that sustain chronic inflammation [88].
Studies show that olive oil-based diets improve PI healing by reducing inflammation, oxidative stress, and enhancing collagen synthesis [89]. Anti-inflammatory diets have proven beneficial for SCI patients [90].
An optimal SCI diet should: 1) reduce inflammation and oxidative stress, 2) enhance immunity, 3) support gut microbiota, and 4) limit AGEs [69, 91]. Key features of such a diet include: 1) high omega-3 content [92], 2) low omega-6/omega-3 ratio [93], 3) high intake of polyphenols and antioxidants from herbs, fruits, and vegetables [94], 4) low glycemic load [95], and 5) minimal ultra-processed foods [96]. While direct links with SCI are unconfirmed, AGEs trigger oxidative stress and inflammation, impair endothelial function and angiogenesis, common issues in chronic illness [97]. Both food content and cooking methods affect AGE levels [98]. Strategies to reduce dietary AGEs include: 1) avoiding high-AGE foods, 2) using cooking methods that limit AGE formation, and 3) reducing intake of AGE-promoting ingredients, such as fructose. While foods rich in AGEs are aged and high-fat cheeses and dry-processed carbohydrates (e.g., crackers), AGE content increases depending on the cooking method (Table 4) [98, 99].
 Click to view | Table 4. Examples to Better Understand How the Cooking Method Can Be Decisive |
Anti-inflammatory compounds, such as quercetin, resveratrol, curcumin, and catechin, can help counteract AGE accumulation [100].
Hydration is essential for wound healing and skin health. Recommended fluid intake is 1 mL per kcal of energy needs plus 500 mL, or 40 mL/kg of body mass plus 500 mL [101]. Additional fluids are needed for those with draining wounds or excessive sweating. Immunonutrition formulas, typically including arginine, glutamine, omega-3s, vitamins, and trace minerals, have been associated with reduced infections and inflammation and improved gut health and wound healing [102]. These dietary principles form the foundation of nutritional strategies for SCI patients with or at risk for PIs (Table 5).
 Click to view | Table 5. Key Dietary Components to Improve Immune Function and Gut Health |
Patient education on precise dietary choices is essential and should be viewed as part of standard medical care. The proposed dietary plan is based on Mediterranean diet, which is consistently showing beneficial effects on chronic conditions [103], and targets an 81-kg tetraplegic patient with no major comorbidities (Tables 6–11). The plan assumes an energy intake of ∼20 kcal/kg body weight and includes 2 L of calcium-rich water (≥ 300 mg/L). Key points include: 1) sugar intake only from whole foods; 2) optimal calcium intake, excluding water contribution; 3) adequate intake of vitamins, minerals, and amino acids; and 4) suboptimal resveratrol levels, which require supplementation (Fig. 2).
 Click to view | Table 6. Breakfast With Whole Oat Porridge With Berries, Flaxseeds, Kefir, and Almonds |
 Click to view | Table 7. Snack With Pear, Almonds, and Green Tea |
 Click to view | Table 8. Lunch With Whole Wheat Pasta With Chickpeas, Artichokes, Red Onion, Chicory; Steamed or Boiled Chicken Breast; Salad of Fresh Spinach, Red Onion, Avocado, Olive Oil, Turmeric, Ginger, and Black Pepper |
 Click to view | Table 9. Snack With Greek Yogurt With Chia Seeds and Green Tea |
 Click to view | Table 10. Dinner With Baked Salmon Fillet With Broccoli, Red Onion, Whole-Grain Bread, Olive Oil, Turmeric, Ginger, and Black Pepper |
 Click to view | Table 11. Total Daily Intakes |
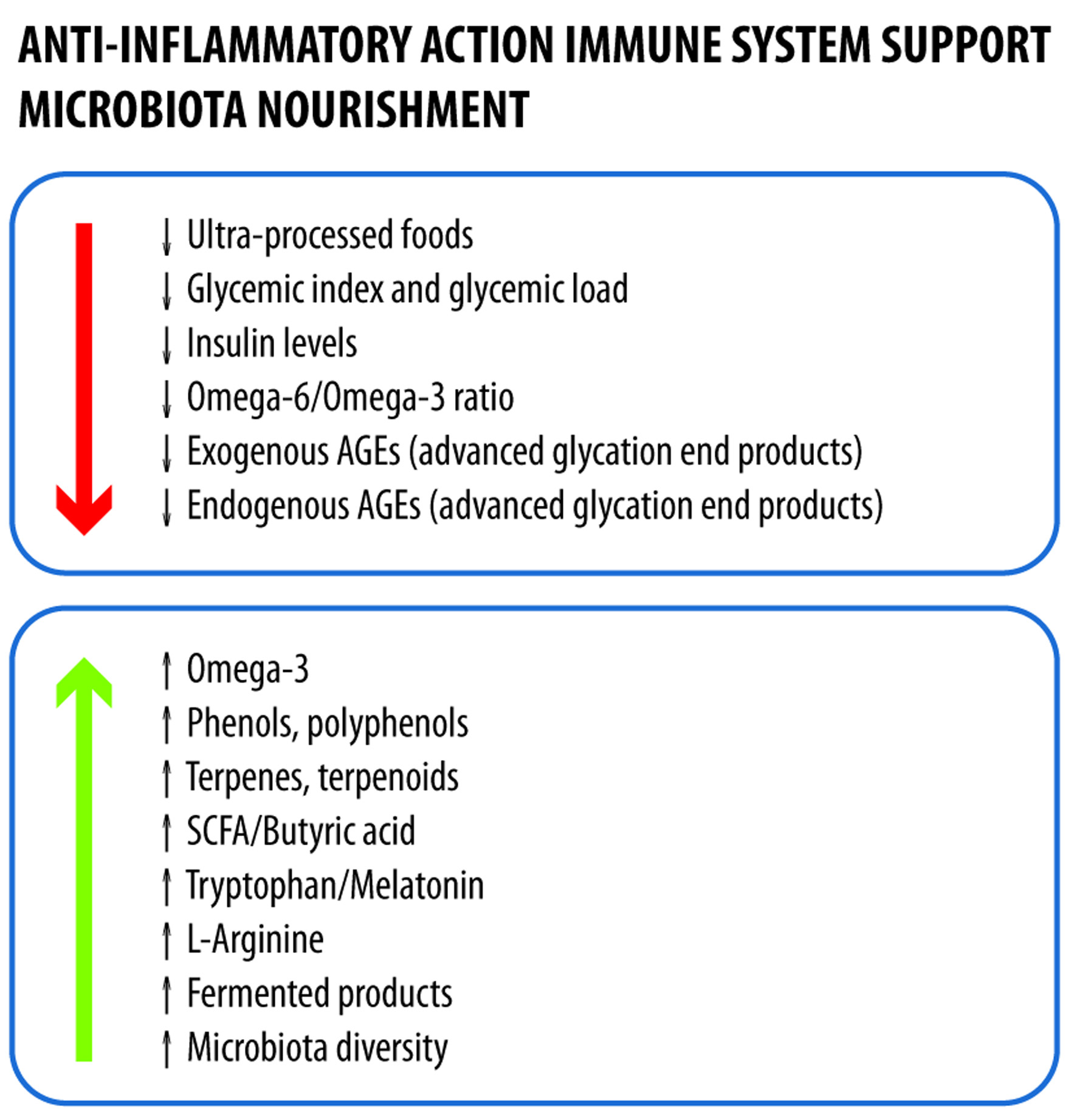 Click for large image | Figure 2. Characteristics of an anti-inflammatory diet that promotes gut health and limits the amount of advanced glycation end products. |
Patient-setting specific management strategies
Effective PI management includes enhancing psychosocial support, ensuring complete pressure relief, optimizing support surfaces and wheelchair seating systems, and providing adequate nutrition. In addition, reducing wound bioburden, eradicating infections, and, when necessary, performing surgical interventions followed by appropriate postoperative care all form essential components of the overall management strategy [104].
Management strategies for the “acute” setting: in-hospital care
In the acute setting, verifying functional outcomes and using pressure redistribution devices are essential [104]. Physical activity helps prevent muscle atrophy, PIs, obesity, inactivity, and bone loss [105]. Patient weight should be assessed to ensure devices fit properly, as standard models may not suit individuals with obesity. For positioning and pressure relief [106], repositioning is advised every 2–4 h with support from nurses or therapists. Wheelchair users should shift weight for at least 30 s every 15 min and reposition hourly [13]. Nutritional support within 72 h is standard for trauma patients. Ongoing clinical and instrumental assessments are needed during acute and post-acute phases, with interventions tailored to high-risk individuals [59]. At discharge, nutrition counseling and assessing awareness of the role of diet in PI prevention and healing are strongly recommended.
Management strategies for the “chronic” setting
In the chronic setting, PI prevention strategies mirror those in acute and post-acute phases and include daily skin checks for early detection, minimizing risk factors, and maintaining a balanced diet with nutritional counseling [107]. The authors emphasized the importance of educating patients and caregivers on PI risk factors and the role of nutrition in skin integrity and healing. To simplify dietary guidance, the authors introduced a Food Suitability Map that uses color-coding to classify foods based on nutritional value, drawing from international literature and databases. Foods are categorized by intake frequency within an anti-inflammatory Mediterranean diet [88, 103, 108]. Foods classified as “green” are those for daily use, with high therapeutic value. Foods classified as “yellow” are those for moderate weekly use with limited benefits. Foods classified as “orange” are those for occasional weekly use and display minimal benefit or mild risk. Foods classified as “red” are those for rare intake or to be avoided, due to potential harm and limited value (Fig. 3).
 Click for large image | Figure 3. A traffic light food map for SCI patients with PIs. PIs: pressure injuries; SCI: spinal cord injury. |
Additional factors, such as bowel and sphincter function, colostomy presence, diabetes, QoL, physical activity, and eating habits, should be evaluated, with a 1-month follow-up recommended.
A food diary is essential for SCI patients to track intake, support metabolic health, PI prevention, and adherence to personalized plans. As biomarkers provide limited insight due to physiological variability and cannot guide dietary change [109], direct food tracking remains the most practical method. The authors advocate for a food diary as an interactive tool involving patients and caregivers, enhanced by the Food Suitability Map. This integration helps identify dietary patterns and necessary modifications. Core features of an effective food diary are listed in Table 12, and the diary templates (Figs 4 and 5) align with the Nutritional Therapeutic Plan. Color-coded entries reflect prescribed meals and guide appropriate substitutions based on the Suitability Map. A sample entry is shown in Figure 5.
 Click to view | Table 12. Key Features of a Food Diary for Spinal Cord Injury Patients |
 Click for large image | Figure 4. Template of a food diary for recording consumption according to the prescribed meal plan for breakfast. |
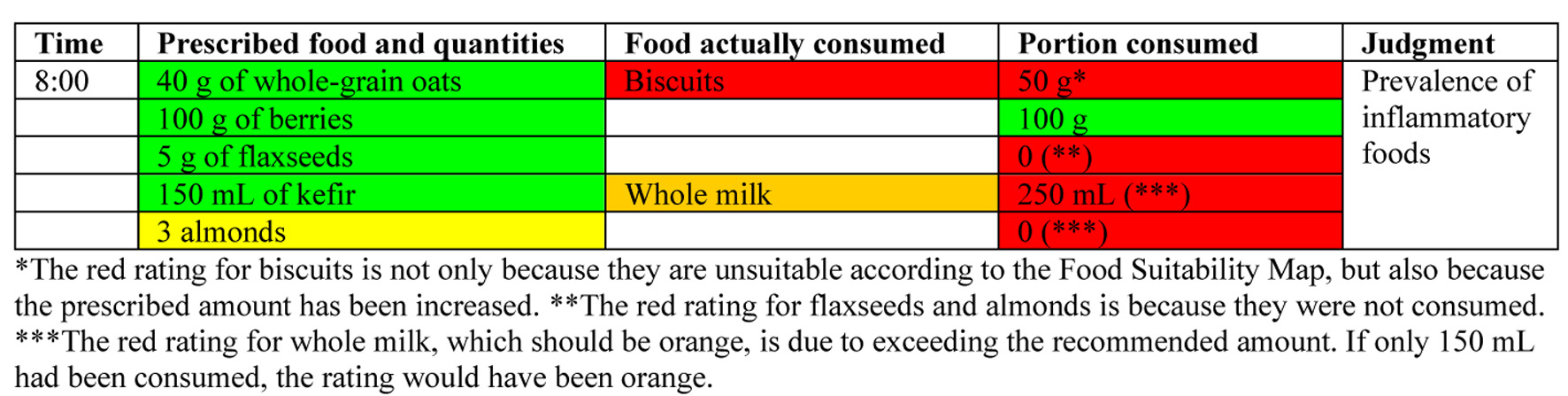 Click for large image | Figure 5. Example of how a food diary could look after patient interaction. |
Management strategies for the “chronic managed by local healthcare services” setting
The authors highlighted the need for professional figures in this setting, as certified specialists in SCI management are often lacking. To address this, the authors suggested periodic follow-ups and specialized training programs to enable HCPs to provide optimal care. The recommendations outlined for other care settings should also be applied in this context.
Management strategies for the “chronic patients readmitted to the hospital with complications” setting
A concerning statistic regards hospital readmissions of chronic SCI, 40% of cases of which are due to PIs. The authors emphasized the need for effective community-level handovers, as spinal units alone cannot meet growing care demands. A structured network between hospitals and local HCPs, based on clear communication, defined roles, and coordinated actions, is crucial for ensuring care continuity. In PI surgery, nutritional interventions should be initiated not only postoperatively [110], but also preoperatively as part of a “prehabilitation” strategy. The authors support incorporating dietary measures into a tailored Enhanced Recovery After Surgery (ERAS) protocol, encompassing pre-, intra-, and postoperative phases to improve recovery, prevent complications, and optimize outcomes [104]. Before PI-related surgery, patients should undergo gastroenterological evaluation to assess colostomy needs. Rehabilitation specialists and occupational therapists (OTs) evaluate home care, seating, pressure relief, and bladder function. Dietitians help tailor nutrition plans to support wound healing and muscle maintenance. OTs also assess skin integrity, identify risks, recommend preventive strategies, evaluate the adequacy of pressure-relief devices, and guide patients and caregivers on skin inspection. Psychologists support emotional well-being and adherence to care plans, enhancing motivation, coping, and long-term self-management [13, 111].
| Conclusions | ▴Top |
Managing patients with SCIs requires a multidisciplinary approach, with nutrition playing a key role in preventing and treating PIs. Individualized dietary strategies are essential to address SCI-related metabolic and inflammatory issues. Supporting gut health with fiber, probiotics, and anti-inflammatory foods alongside the intake of micronutrients (e.g., vitamins A, C, zinc, iron) and adequate hydration can promote wound healing and lower PI risk, helping maintain immune function and reduce inflammation. Personalized dietary plans, guided by qualified dietitians, should meet energy and nutrient needs in SCI. Tools such as food diaries and dietary maps can enhance education, adherence, and informed decision-making. Implementing evidence-based nutrition protocols can improve outcomes, reduce costs, and enhance QoL for SCI patients. Future research should refine dietary interventions, evaluate nutraceuticals, and assess the long-term impact of nutrition on PI prevention and healing.
Acknowledgments
The authors acknowledge Lara Vecchi, Aashni Shah, and Valentina Attanasio (Polistudium Srl, Milan, Italy) for medical writing, editorial assistance, and English language editing. Coordination was provided by Francesca Cappellini, PhD (Polistudium SRL, Milan, Italy).
Financial Disclosure
The medical writing was unconditionally funded by B. Braun SpA.
Conflict of Interest
AC is an employee of B. Braun. GDP, LDP, AA, PV, WC and LP declare no conflict of interest.
Author Contributions
All authors contributed to the discussions that formed the basis of this expert opinion. The clinicians (AA, WC, AC, GDP, LDP, and LP) identified and reviewed the literature relevant to their respective areas of expertise in spinal cord injury management, wound care, rehabilitation, internal medicine, and urology. PV, as the nutritionist, provided specific input on nutritional strategies for the prevention and management of skin lesions, including evidence-based dietary interventions. All authors contributed to the manuscript drafting, critically revised the manuscript and approved the final version for submission.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- Kirshblum SC, Biering-Sorensen F, Betz R, Burns S, Donovan W, Graves DE, Johansen M, et al. International standards for neurological classification of spinal cord injury: cases with classification challenges. Top Spinal Cord Inj Rehabil. 2014;20(2):81-89.
doi pubmed - Lima R, Monteiro A, Salgado AJ, Monteiro S, Silva NA. Pathophysiology and therapeutic approaches for spinal cord injury. Int J Mol Sci. 2022;23(22):13833.
doi pubmed - Margetis K, Das JM, Emmady PD. Spinal cord injuries. In: StatPearls. Treasure Island (FL) with ineligible companies. 2025.
pubmed - Bach JR. Noninvasive respiratory management of high level spinal cord injury. J Spinal Cord Med. 2012;35(2):72-80.
doi pubmed - Foran SJ, Taran S, Singh JM, Kutsogiannis DJ, McCredie V. Timing of tracheostomy in acute traumatic spinal cord injury: A systematic review and meta-analysis. J Trauma Acute Care Surg. 2022;92(1):223-231.
doi pubmed - Groah SL, Schladen M, Pineda CG, Hsieh CH. Prevention of pressure ulcers among people with spinal cord injury: a systematic review. PM R. 2015;7(6):613-636.
doi pubmed - Wong S, Derry F, Jamous A, Hirani SP, Grimble G, Forbes A. The prevalence of malnutrition in spinal cord injuries patients: a UK multicentre study. Br J Nutr. 2012;108(5):918-923.
doi pubmed - Farkas GJ, Gater DR. Energy expenditure and nutrition in neurogenic obesity following spinal cord injury. J Phys Med Rehabil. 2020;2(1):11-13.
pubmed - Farkas GJ, Sneij A, McMillan DW, Tiozzo E, Nash MS, Gater DR, Jr. Energy expenditure and nutrient intake after spinal cord injury: a comprehensive review and practical recommendations. Br J Nutr. 2022;128(5):863-887.
doi pubmed - Valido E, Bertolo A, Frankl GP, Itodo OA, Pinheiro T, Pannek J, Kopp-Heim D, et al. Systematic review of the changes in the microbiome following spinal cord injury: animal and human evidence. Spinal Cord. 2022;60(4):288-300.
doi pubmed - Thibault-Halman G, Casha S, Singer S, Christie S. Acute management of nutritional demands after spinal cord injury. J Neurotrauma. 2011;28(8):1497-1507.
doi pubmed - Zeng X, Xiang J, Dong L, Dong B, Sun Y. Advance in nutritional status and intervention after spinal cord injury. J Clin Nurs Res 2021;5:7-14.
doi - European pressure ulcer advisory panel, national pressure injury advisory panel and pan pacific pressure injury alliance. Prevention and treatment of pressure ulcers/injuries: clinical practice guideline. The international guideline. Emily Haesler (Ed.). EPUAP/NPIAP/PPPIA: 2019. https://static1.squarespace.com/static/6479484083027f25a6246fcb/t/6553d3440e18d57a550c4e7e/1699992399539/CPG2019edition-digital-Nov2023version.pdf.
- Brienza D, Krishnan S, Karg P, Sowa G, Allegretti AL. Predictors of pressure ulcer incidence following traumatic spinal cord injury: a secondary analysis of a prospective longitudinal study. Spinal Cord. 2018;56(1):28-34.
doi pubmed - Stillman MD, Barber J, Burns S, Williams S, Hoffman JM. Complications of spinal cord injury over the first year after discharge from inpatient rehabilitation. Arch Phys Med Rehabil. 2017;98(9):1800-1805.
doi pubmed - Chen HL, Cai JY, Du L, Shen HW, Yu HR, Song YP, Zha ML. Incidence of pressure injury in individuals with spinal cord injury: a systematic review and meta-analysis. J Wound Ostomy Continence Nurs. 2020;47(3):215-223.
doi pubmed - Flury I, Mueller G, Perret C. The risk of malnutrition in patients with spinal cord injury during inpatient rehabilitation-A longitudinal cohort study. Front Nutr. 2023;10:1085638.
doi pubmed - Areni A, Capeci W, Cassinis A, De Palma L, Del Popolo G, Fergnani F, Pelizzari L. What we do and what we should do against malnutrition in spinal cord injury: A position paper from Italian spinal cord injury network rehabilitation centers. J Clin Med Res. 2024;16(4):138-154.
doi pubmed - Chen B, Yang Y, Cai F, Zhu C, Lin S, Huang P, Zhang L. Nutritional status as a predictor of the incidence of pressure injury in adults: A systematic review and meta-analysis. J Tissue Viability. 2023;32(3):339-348.
doi pubmed - Krause JS, Saunders LL. Health, secondary conditions, and life expectancy after spinal cord injury. Arch Phys Med Rehabil. 2011;92(11):1770-1775.
doi pubmed - Ghaisas S, Pyatak EA, Blanche E, Blanchard J, Clark F, PUPS II Study Group. Lifestyle changes and pressure ulcer prevention in adults with spinal cord injury in the pressure ulcer prevention study lifestyle intervention. Am J Occup Ther. 2015;69(1):6901290020p1-10.
doi pubmed - Gorecki C, Brown JM, Nelson EA, Briggs M, Schoonhoven L, Dealey C, Defloor T, et al. Impact of pressure ulcers on quality of life in older patients: a systematic review. J Am Geriatr Soc. 2009;57(7):1175-1183.
doi pubmed - Guest JF, Fuller GW, Vowden P. Cohort study evaluating the burden of wounds to the UK's National Health Service in 2017/2018: update from 2012/2013. BMJ Open. 2020;10(12):e045253.
doi pubmed - Tate DG, Kalpakjian CZ, Forchheimer MB. Quality of life issues in individuals with spinal cord injury. Arch Phys Med Rehabil. 2002;83(12 Suppl 2):S18-25.
doi pubmed - Le Fort M, Espagnacq M, Albert T, Lefevre C, Perrouin-Verbe B, Ravaud JF. Risk of pressure ulcers in tetraplegic people: a French survey crossing regional experience with a long-term follow-up. Eur J Public Health. 2018;28(6):993-999.
doi pubmed - Perez-Lopez S, Perez-Basterrechea M, Garcia-Gala JM, Martinez-Revuelta E, Fernandez-Rodriguez A, Alvarez-Viejo M. Stem cell and tissue engineering approaches in pressure ulcer treatment. J Spinal Cord Med. 2023;46(2):194-203.
doi pubmed - Brienza D, Kelsey S, Karg P, Allegretti A, Olson M, Schmeler M, Zanca J, et al. A randomized clinical trial on preventing pressure ulcers with wheelchair seat cushions. J Am Geriatr Soc. 2010;58(12):2308-2314.
doi pubmed - Tervo-Heikkinen T, Heikkila A, Koivunen M, Kortteisto T, Peltokoski J, Salmela S, Sankelo M, et al. Nursing interventions in preventing pressure injuries in acute inpatient care: a cross-sectional national study. BMC Nurs. 2023;22(1):198.
doi pubmed - Seth I, Lim B, Cevik J, Gracias D, Chua M, Kenney PS, Rozen WM, et al. Impact of nutrition on skin wound healing and aesthetic outcomes: A comprehensive narrative review. JPRAS Open. 2024;39:291-302.
doi pubmed - Kottner J, Cuddigan J, Carville K, Balzer K, Berlowitz D, Law S, Litchford M, et al. Prevention and treatment of pressure ulcers/injuries: The protocol for the second update of the international Clinical Practice Guideline 2019. J Tissue Viability. 2019;28(2):51-58.
doi pubmed - Salzberg CA, Byrne DW, Cayten CG, van Niewerburgh P, Murphy JG, Viehbeck M. A new pressure ulcer risk assessment scale for individuals with spinal cord injury. Am J Phys Med Rehabil. 1996;75(2):96-104.
doi pubmed - Wilczweski P, Grimm D, Gianakis A, Gill B, Sarver W, McNett M. Risk factors associated with pressure ulcer development in critically ill traumatic spinal cord injury patients. J Trauma Nurs. 2012;19(1):5-10.
doi pubmed - Truglio-Londrigan M, Slyer JT. Shared decision-making for nursing practice: an integrative review. Open Nurs J. 2018;12:1-14.
doi pubmed - Shiferaw WS, Akalu TY, Mulugeta H, Aynalem YA. The global burden of pressure ulcers among patients with spinal cord injury: a systematic review and meta-analysis. BMC Musculoskelet Disord. 2020;21(1):334.
doi pubmed - Sadeghi Fazel F, Derakhshanrad N, Yekaninejad MS, Vosoughi F, Derakhshanrad A, Saberi H. Predictive value of braden risk factors in pressure ulcers of outpatients with spinal cord injury. Acta Med Iran. 2018;56(1):56-61.
pubmed - Giovannoni L, Longobucco Y, Iovino P, Barbetti C, Becattini S, Bonanni D, Cordelli F, et al. Complementing Braden scale for pressure ulcer risk with clinical and demographic-related factors in a large cohort of hospitalized Italian patients. J Tissue Viability. 2024;33(2):243-247.
doi pubmed - Afridi A, Rathore FA. Are risk assessment tools effective for the prevention of pressure ulcers formation? A cochrane review summary with commentary. Am J Phys Med Rehabil. 2020;99(4):357-358.
doi pubmed - Flett HM, Delparte JJ, Scovil CY, Higgins J, Laramee MT, Burns AS. Determining pressure injury risk on admission to inpatient spinal cord injury rehabilitation: a comparison of the FIM, spinal cord injury pressure ulcer scale, and braden scale. Arch Phys Med Rehabil. 2019;100(10):1881-1887.
doi pubmed - Zanotti R, Segala M, Bovo A. Validation of LPD scale for the assessment of pressure ulcer at home. Open J Nurs. 2023;13:267-282.
doi - Ledger LJ, Worsley PR, Hope J, Schoonhoven L. Patient perceptions and understanding of pressure ulcer risk in the community: Empirical Research Qualitative. J Adv Nurs. 2023;79(9):3312-3323.
doi pubmed - Black JM, Berke CT. Deep tissue pressure injuries: identification, treatment, and outcomes among critical care patients. Crit Care Nurs Clin North Am. 2020;32(4):563-572.
doi pubmed - Angel DE, Lloyd P, Carville K, Santamaria N. The clinical efficacy of two semi-quantitative wound-swabbing techniques in identifying the causative organism(s) in infected cutaneous wounds. Int Wound J. 2011;8(2):176-185.
doi pubmed - Sgarzani R, Rucci P, Landi S, Battilana M, Capirossi R, Aramini B, Negosanti L. Reconstructive surgery of pressure injuries in spinal cord injury/disorder patients: retrospective observational study and proposal of an algorithm for the flap choice. Healthcare (Basel). 2023;12(1):34.
doi pubmed - Farkas GJ, Pitot MA, Berg AS, Gater DR. Nutritional status in chronic spinal cord injury: a systematic review and meta-analysis. Spinal Cord. 2019;57(1):3-17.
doi pubmed - Keller U. Nutritional laboratory markers in malnutrition. J Clin Med. 2019;8(6):775.
doi pubmed - Nightingale TE, Gorgey AS. Predicting basal metabolic rate in men with motor complete spinal cord injury. Med Sci Sports Exerc. 2018;50(6):1305-1312.
doi pubmed - Gater DR, Jr., Farkas GJ, Tiozzo E. Pathophysiology of neurogenic obesity after spinal cord injury. Top Spinal Cord Inj Rehabil. 2021;27(1):1-10.
doi pubmed - Cleland DA, Eranki AP. Procalcitonin. In: StatPearls. Treasure Island (FL) ineligible companies. 2025.
pubmed - Bazzocchi G, Turroni S, Bulzamini MC, D'Amico F, Bava A, Castiglioni M, Cagnetta V, et al. Changes in gut microbiota in the acute phase after spinal cord injury correlate with severity of the lesion. Sci Rep. 2021;11(1):12743.
doi pubmed - Tulsky DS, Kisala PA. The Spinal Cord Injury—Quality of Life (SCI-QOL) measurement system: Development, psychometrics, and item bank calibration. J Spinal Cord Med. 2015;38(3):251-256.
doi pubmed - Development of the World Health Organization WHOQOL-BREF quality of life assessment. The WHOQOL Group. Psychol Med. 1998;28(3):551-558.
doi pubmed - Barchitta M, Maugeri A, Favara G, Magnano San Lio R, Evola G, Agodi A, Basile G. Nutrition and wound healing: an overview focusing on the beneficial effects of curcumin. Int J Mol Sci. 2019;20(5):1119.
doi pubmed - Munoz N, Posthauer ME, Cereda E, Schols J, Haesler E. The role of nutrition for pressure injury prevention and healing: the 2019 international clinical practice guideline recommendations. Adv Skin Wound Care. 2020;33(3):123-136.
doi pubmed - Dorner B, Posthauer ME, Thomas D, National Pressure Ulcer Advisory P. The role of nutrition in pressure ulcer prevention and treatment: National Pressure Ulcer Advisory Panel white paper. Adv Skin Wound Care. 2009;22(5):212-221.
doi pubmed - Cox J, Rasmussen L. Enteral nutrition in the prevention and treatment of pressure ulcers in adult critical care patients. Crit Care Nurse. 2014;34(6):15-27.
doi pubmed - Saghaleini SH, Dehghan K, Shadvar K, Sanaie S, Mahmoodpoor A, Ostadi Z. Pressure ulcer and nutrition. Indian J Crit Care Med. 2018;22(4):283-289.
doi pubmed - Moughan PJ, Lim WXJ. Digestible indispensable amino acid score (DIAAS): 10 years on. Front Nutr. 2024;11:1389719.
doi pubmed - Matthews JJ, Arentson-Lantz EJ, Moughan PJ, Wolfe RR, Ferrando AA, Church DD. Understanding dietary protein quality: digestible indispensable amino acid scores and beyond. J Nutr. 2025;155(10):3152-3167.
doi pubmed - https://spinalis.se/wp-content/uploads/2019/05/Spinalis_broschyr_eng_2019_web-1.pdf.
- Groah SL, Nash MS, Ljungberg IH, Libin A, Hamm LF, Ward E, Burns PA, et al. Nutrient intake and body habitus after spinal cord injury: an analysis by sex and level of injury. J Spinal Cord Med. 2009;32(1):25-33.
doi pubmed - Sabour H, Nazari M, Latifi S, Soltani Z, Shakeri H, Larijani B, Ghodsi SM, et al. The relationship between dietary intakes of amino acids and bone mineral density among individuals with spinal cord injury. Oman Med J. 2016;31(1):22-28.
doi pubmed - Academy of Nutrition and Dietetics. Spinal Cord Injury (SCI) guidelines Chicago, Illinois. 2009. https://andeal.org/topic.cfm? menu=5292&pcat=3487&cat=5448. Last Access May 25, 2023.
- Wong S, Derry F, Jamous A, Hirani SP, Grimble G, Forbes A. Validation of the spinal nutrition screening tool (SNST) in patients with spinal cord injuries (SCI): result from a multicentre study. Eur J Clin Nutr. 2012;66(3):382-387.
doi pubmed - Nash MS, Groah SL, Gater DR, Jr., Dyson-Hudson TA, Lieberman JA, Myers J, Sabharwal S, et al. Identification and management of cardiometabolic risk after spinal cord injury: clinical practice guideline for health care providers. Top Spinal Cord Inj Rehabil. 2018;24(4):379-423.
doi pubmed - Paralyzed veterans of america: Washington, DC, USA; Academy of nutrition and dietetics. Spinal Cord Injury (SCI) Guidelines Chicago, Illinois. 2009. https://andeal.org/topic.cfm?menu=5292&pcat=3487&cat=5448. Last Access May 25, 2023.
- Pellegrini CA, Burkhart L, Jones K, LaVela SL. Health provider identified barriers and facilitators to weight management for individuals with spinal cord injury. Spinal Cord. 2021;59(10):1061-1071.
doi pubmed - Langer G, Wan CS, Fink A, Schwingshackl L, Schoberer D. Nutritional interventions for preventing and treating pressure ulcers. Cochrane Database Syst Rev. 2024;2(2):CD003216.
doi pubmed - Schwab JM, Zhang Y, Kopp MA, Brommer B, Popovich PG. The paradox of chronic neuroinflammation, systemic immune suppression, autoimmunity after traumatic chronic spinal cord injury. Exp Neurol. 2014;258:121-129.
doi pubmed - Kellow NJ, Coughlan MT. Effect of diet-derived advanced glycation end products on inflammation. Nutr Rev. 2015;73(11):737-759.
doi pubmed - Doi T, Inoue T, Sugaya J, Horii C, Tozawa K, Nakarai H, Sasaki K, et al. Noninvasive skin autofluorescence of advanced glycation end products in patients with degenerative cervical myelopathy. World Neurosurg. 2025;194:123556.
doi pubmed - Palego L, Betti L, Rossi A, Giannaccini G. Tryptophan biochemistry: structural, nutritional, metabolic, and medical aspects in humans. J Amino Acids. 2016;2016:8952520.
doi pubmed - Platten M, Nollen EAA, Rohrig UF, Fallarino F, Opitz CA. Tryptophan metabolism as a common therapeutic target in cancer, neurodegeneration and beyond. Nat Rev Drug Discov. 2019;18(5):379-401.
doi pubmed - Taleb S. Tryptophan dietary impacts gut barrier and metabolic diseases. Front Immunol. 2019;10:2113.
doi pubmed - Tomita H, Egashira K, Ohara Y, Takemoto M, Koyanagi M, Katoh M, Yamamoto H, et al. Early induction of transforming growth factor-beta via angiotensin II type 1 receptors contributes to cardiac fibrosis induced by long-term blockade of nitric oxide synthesis in rats. Hypertension. 1998;32(2):273-279.
doi pubmed - Cani PD, Amar J, Iglesias MA, Poggi M, Knauf C, Bastelica D, Neyrinck AM, et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56(7):1761-1772.
doi pubmed - Neves AL, Coelho J, Couto L, Leite-Moreira A, Roncon-Albuquerque R, Jr. Metabolic endotoxemia: a molecular link between obesity and cardiovascular risk. J Mol Endocrinol. 2013;51(2):R51-64.
doi pubmed - Kigerl KA, Hall JC, Wang L, Mo X, Yu Z, Popovich PG. Gut dysbiosis impairs recovery after spinal cord injury. J Exp Med. 2016;213(12):2603-2620.
doi pubmed - Liu J, An H, Jiang D, Huang W, Zou H, Meng C, Li H. Study of bacterial translocation from gut after paraplegia caused by spinal cord injury in rats. Spine (Phila Pa 1976). 2004;29(2):164-169.
doi pubmed - Medeiros BA, dos Santos CL, Palheta RC, Jr., de Queiroz DA, da Graca JR, dos Santos AA, Rola FH, et al. Spinal cord transection modifies ileal fluid and electrolyte transport in rats. Auton Neurosci. 2008;139(1-2):24-29.
doi pubmed - Puddu A, Sanguineti R, Montecucco F, Viviani GL. Evidence for the gut microbiota short-chain fatty acids as key pathophysiological molecules improving diabetes. Mediators Inflamm. 2014;2014:162021.
doi pubmed - Zhang L, Dong D, Jiang C, Li Z, Wang X, Peng Y. Insight into alteration of gut microbiota in Clostridium difficile infection and asymptomatic C. difficile colonization. Anaerobe. 2015;34:1-7.
doi pubmed - Zhao J, Nian L, Kwok LY, Sun T, Zhao J. Reduction in fecal microbiota diversity and short-chain fatty acid producers in Methicillin-resistant Staphylococcus aureus infected individuals as revealed by PacBio single molecule, real-time sequencing technology. Eur J Clin Microbiol Infect Dis. 2017;36(8):1463-1472.
doi pubmed - Wang M, Wichienchot S, He X, Fu X, Huang Q, Zhang B. In vitro colonic fermentation of dietary fibers: Fermentation rate, short-chain fatty acid production and changes in microbiota. Trends Food Sci Technol. 2019;88:1-9.
doi - Le Chatelier E, Nielsen T, Qin J, Prifti E, Hildebrand F, Falony G, Almeida M, et al. Richness of human gut microbiome correlates with metabolic markers. Nature. 2013;500(7464):541-546.
doi pubmed - Xie L, Wu H, Huang X, Yu T. Melatonin, a natural antioxidant therapy in spinal cord injury. Front Cell Dev Biol. 2023;11:1218553.
doi pubmed - Barrett E, Ross RP, O'Toole PW, Fitzgerald GF, Stanton C. gamma-Aminobutyric acid production by culturable bacteria from the human intestine. J Appl Microbiol. 2012;113(2):411-417.
doi pubmed - Bloomfield HE, Kane R, Koeller E, Greer N, MacDonald R, Wilt T. In: Benefits and harms of the mediterranean diet compared to other diets. Washington (DC), 2015.
pubmed - Yu X, Pu H, Voss M. Overview of anti-inflammatory diets and their promising effects on non-communicable diseases. Br J Nutr. 2024;132(7):898-918.
doi pubmed - Schanuel FS, Saguie BO, Monte-Alto-Costa A. Olive oil promotes wound healing of mice pressure injuries through NOS-2 and Nrf2. Appl Physiol Nutr Metab. 2019;44(11):1199-1208.
doi pubmed - Allison DJ, Beaudry KM, Thomas AM, Josse AR, Ditor DS. Changes in nutrient intake and inflammation following an anti-inflammatory diet in spinal cord injury. J Spinal Cord Med. 2019;42(6):768-777.
doi pubmed - Van Puyvelde K, Mets T, Njemini R, Beyer I, Bautmans I. Effect of advanced glycation end product intake on inflammation and aging: a systematic review. Nutr Rev. 2014;72(10):638-650.
doi pubmed - Calder PC. Omega-3 fatty acids and inflammatory processes: from molecules to man. Biochem Soc Trans. 2017;45(5):1105-1115.
doi pubmed - DiNicolantonio JJ, O'Keefe J. The importance of maintaining a low omega-6/omega-3 ratio for reducing the risk of autoimmune diseases, asthma, and allergies. Mo Med. 2021;118(5):453-459.
pubmed - Stromsnes K, Correas AG, Lehmann J, Gambini J, Olaso-Gonzalez G. Anti-inflammatory properties of diet: role in healthy aging. Biomedicines. 2021;9(8):922.
doi pubmed - Sears B. Anti-inflammatory diets. J Am Coll Nutr. 2015;34(Suppl 1):14-21.
doi pubmed - Tristan Asensi M, Napoletano A, Sofi F, Dinu M. Low-grade inflammation and ultra-processed foods consumption: a review. Nutrients. 2023;15(6):1546.
doi pubmed - Liu J, Pan S, Wang X, Liu Z, Zhang Y. Role of advanced glycation end products in diabetic vascular injury: molecular mechanisms and therapeutic perspectives. Eur J Med Res. 2023;28(1):553.
doi pubmed - Uribarri J, Woodruff S, Goodman S, Cai W, Chen X, Pyzik R, Yong A, et al. Advanced glycation end products in foods and a practical guide to their reduction in the diet. J Am Diet Assoc. 2010;110(6):911-916.e912.
doi pubmed - Story M, Hayes M, Kalina B. Availability of foods in high schools: is there cause for concern? J Am Diet Assoc. 1996;96(2):123-126.
doi pubmed - Zawada A, Machowiak A, Rychter AM, Ratajczak AE, Szymczak-Tomczak A, Dobrowolska A, Krela-Kazmierczak I. Accumulation of advanced glycation end-products in the body and dietary habits. Nutrients. 2022;14(19):3982.
doi pubmed - Bernardi M, Fedullo AL, Bernardi E, Munzi D, Peluso I, Myers J, Lista FR, et al. Diet in neurogenic bowel management: a viewpoint on spinal cord injury. World J Gastroenterol. 2020;26(20):2479-2497.
doi pubmed - Chow O, Barbul A. Immunonutrition: role in wound healing and tissue regeneration. Adv Wound Care (New Rochelle). 2014;3(1):46-53.
doi pubmed - Finicelli M, Di Salle A, Galderisi U, Peluso G. The mediterranean diet: an update of the clinical trials. Nutrients. 2022;14(14):2956.
doi pubmed - Melnyk M, Casey RG, Black P, Koupparis AJ. Enhanced recovery after surgery (ERAS) protocols: Time to change practice? Can Urol Assoc J. 2011;5(5):342-348.
doi pubmed - Chen SC, Lai CH, Chan WP, Huang MH, Tsai HW, Chen JJ. Increases in bone mineral density after functional electrical stimulation cycling exercises in spinal cord injured patients. Disabil Rehabil. 2005;27(22):1337-1341.
doi pubmed - Team V, Bouguettaya A, Richards C, Turnour L, Jones A, Teede H, Weller CD. Patient education materials on pressure injury prevention in hospitals and health services in Victoria, Australia: Availability and content analysis. Int Wound J. 2020;17(2):370-379.
doi pubmed - Caliri MH. Spinal cord injury and pressure ulcers. Nurs Clin North Am. 2005;40(2):337-347.
doi pubmed - O'Keefe JH, Torres-Acosta N, O'Keefe EL, Saeed IM, Lavie CJ, Smith SE, Ros E. A pesco-mediterranean diet with intermittent fasting: JACC review topic of the week. J Am Coll Cardiol. 2020;76(12):1484-1493.
doi pubmed - Wild CP, Andersson C, O'Brien NM, Wilson L, Woods JA. A critical evaluation of the application of biomarkers in epidemiological studies on diet and health. Br J Nutr. 2001;86(Suppl 1):S37-53.
doi pubmed - Chu Y, Kong J, Xu J, Han G, Yu W, Xu X. Role of nutritional support in nursing practice for improving surgical site wound healing in patients post-surgery with risk of pressure ulcers. Int Wound J. 2024;21(4):e14855.
doi pubmed - Consortium for Spinal Cord Medicine Clinical Practice G. Pressure ulcer prevention and treatment following spinal cord injury: a clinical practice guideline for health-care professionals. J Spinal Cord Med. 2001;24(Suppl 1):S40-101.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Clinical Medicine Research is published by Elmer Press Inc.