| Journal of Clinical Medicine Research, ISSN 1918-3003 print, 1918-3011 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Med Res and Elmer Press Inc |
| Journal website https://jocmr.elmerjournals.com |
Original Article
Volume 18, Number 4, April 2026, pages 234-242
Usefulness of Blood Rheology as a Predictor of Primary Cardiovascular Disease Events in Patients With Stage G3 Chronic Kidney Disease
Takashi Hitsumoto
Hitsumoto Medical Clinic, 2-7-7, Takezakicyou, Shimonoseki City, Yamaguchi 750-0025, Japan
Manuscript submitted February 18, 2026, accepted March 31, 2026, published online April 15, 2026
Short title: WBPT and Primary CVD Events in Stage G3 CKD
doi: https://doi.org/10.14740/jocmr6541
| Abstract | ▴Top |
Background: Several studies have highlighted that impairment of blood rheology plays a crucial role in the development of cardiovascular disease (CVD) and arteriosclerosis. However, the value of blood rheology as a potential predictor of the development of CVD in patients with chronic kidney disease (CKD) remains unknown. The objective of this prospective study was to investigate the utility of blood rheology using whole blood passage time (WBPT) as a predictor of primary CVD events in patients with stage G3 CKD, which is often encountered in daily practice.
Methods: This study involved 417 outpatients with stage G3 CKD (male/female: 144/273) without history of CVD. WBPT was measured by microchannel array flow analyzer as a commercial device. Subsequently, the author evaluated the effectiveness of WBPT for the prediction of primary CVD events, which were defined as major adverse cardiovascular events (MACEs, i.e., cardiovascular death, nonfatal ischemic heart disease, and non-fatal ischemic stroke).
Results: Patients were assigned into two groups according to the WBPT cut-off value, which was estimated by receiver operating characteristic curve analysis: low (group L, WBPT ≤ 74.3 s; n = 255) or high (group H, WBPT > 74.3 s; n = 162). During the median follow-up period of 76 months (range: 2–96 months), MACEs occurred in 74 patients (group L: n = 17 (6.7%) vs. group H: n = 57 (35.2%); P < 0.001, log-rank test). Multivariate Cox regression analysis revealed that patients in group H were at a significantly higher risk of developing MACEs than those in group L (hazard ratio: 3.89; 95% confidence interval: 2.12–7.10; P < 0.001).
Conclusions: The present findings showed that impairment of blood rheology, determined using WBPT, is predictive of primary CVD events in patients with stage G3 CKD. Further large-scale studies are warranted to confirm whether various interventions can reduce the incidence of primary CVD events with improved WBPT in patients with stage G3 CKD.
Keywords: Blood rheology; Whole blood passage time; Chronic kidney disease stage G3; Cardiovascular events; Augmentation index; Skin autofluorescence; Renin–angiotensin system inhibitor; Prospective study
| Introduction | ▴Top |
Several studies have highlighted that impairment of blood rheology plays an important role in the development of cardiovascular disease (CVD) and arteriosclerosis [1, 2]. Numerous methods are used for the evaluation of blood rheology; among those, a commercially available device using microscope images, termed microchannel array flow analyzer (MC-FAN), has been utilized for the measurement of whole blood passage time (WBPT) [3]. The MC-FAN assesses blood rheology through the flow of the patient’s whole blood into an artificial vascular model. It has been shown that this method is simple and reliable; moreover, clinical evidence has demonstrated the significance of WBPT as an indicator of the risk of developing CVD [4–8].
In recent years, the incidence of chronic kidney disease (CKD) has been increasing worldwide due to the aging population and increase in lifestyle-related diseases (e.g., diabetes mellitus, dyslipidemia, and hypertension) [9, 10]. Additionally, several studies reported that CKD is closely associated with the incidence of CVD [11, 12]. An estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m2 is indicative of stages G3–5 CKD. In clinical practice, the number of patients with stage G3 CKD is markedly higher than that of patients with stage G4 or G5 disease [13, 14]. Conversely, previous studies have reported that the incidence of CVD events in CKD in the G4–5 period is high [15]. Therefore, it would be clinically meaningful if we could detect high-risk cases of CVD corresponding to stage G4–5 CKD in patients with stage G3 CKD, which are often encountered in daily practice. The aim of this prospective study was to investigate the clinical utility of blood rheology using WBPT as a predictor of primary CVD events in patients with stage G3 CKD.
| Materials and Methods | ▴Top |
Patients
This study is a prospective study at a single center unit. WBPT measurement was blinded and also performed standardized across patients. From July 2017 to June 2019, 450 consecutive stage G3 CKD patients without history of CVD visited the Hitsumoto Medical Clinic, Yamaguchi Prefecture, Japan. Of those, 29 patients who lacked baseline clinical data including WBPT and four patients who failed to provide consent for their participation in this study were excluded. Finally, 417 outpatients (144 males (34.5%) and 273 females (65.5%)) were prospectively enrolled. The eGFR was measured using a formula developed for the Japanese population [16]. For patient classification, an eGFR ranging 30–59 mL/min/1.73 m2 denoted stage G3 CKD.
Ethical considerations
The study protocol was approved by the Institutional Review Board of Hitsumoto Medical Clinic (date of approval: June 14, 2017; Approval Number: HMC-2017-7). The clinical study was conducted in compliance with the ethical principles of the Declaration of Helsinki, which is the ethical principle of medical research involving human beings.
Evaluation of blood rheology
Blood rheology was assessed by measuring the WBPT using a commercialized MC-FAN HR300 rheometer (MC Healthcare Inc., Tokyo, Japan), as previously described [3, 7]. WBPT measurements were performed on the morning of the same day as blood collection by healthcare professionals who were not informed of the study’s content beforehand. The microchannel transit time of saline (100 µL) was initially measured as a control. Subsequently, similar measurements were performed on whole blood heparinized samples (100 µL) obtained from patients. The WBPT measurements were normalized to those of the saline transit time and were measured within 60 min following sample collection. The configuration of the microchannels was as follows: length, 30 µm; width, 7 µm; and depth, 4.5 µm. The reliability of the present WBPT measurements was better than previously reported (i.e., inter- and intra-assay coefficients of variation: 8% and 5%, respectively) [5].
Evaluation of clinical parameters
The degree of obesity was assessed by body mass index, and patients with 25.0 kg/m2 or more were considered obesity [17]. Other CVD risk factors such as smoking, hypertension, dyslipidemia, and diabetes mellitus have been decided using previous report [8]. Blood or urine collection was collected after fasting for more than 12 h. Hematocrit, serum creatinine levels, high-sensitivity C-reactive protein levels, serum lipid levels, blood glucose levels, and urinary albumin levels were measured using standard methods. Albuminuria was defined as ≥ 30 mg/g Cr of urinary albumin levels. Skin autofluorescence (AF) is known to be a marker of advanced glycation end products (AGEs) in vivo; in this analysis, it was measured on the medial forearm of the patients using commercially available devices (AGE Reader; DiagnOptics, Groningen, Netherlands) in a manner similar to previous reports [18, 19]. The radial augmentation index (r-AIx) (marker of arterial reflected waves) was calculated using a tonometry-based device (HEM-9010AI, OMRON Healthcare Co., Ltd., Kyoto, Japan), as in other studies [20, 21]. Oral medications, such as renin–angiotensin system (RAS) inhibitor statin, and anti-diabetes medications were also investigated.
Statistical analysis
The commercialized software Stat View-J 5.0 (HULINKS Inc., Tokyo, Japan) and MedCalc (MedCalc Software, Ostend, Belgium) were used for the statistical analysis. Receiver operating characteristic curves were created, and the maximum Youden index was determined [22]. Continuous variables were expressed as means and standard deviations or median (interquartile range). Student’s t-test or Mann–Whitney U test was used to perform comparisons between groups. Kaplan–Meier analysis was conducted to produce event-free survival curves; differences between these curves were assessed using a log-rank test. Multivariate analysis was performed using multivariate Cox regression analysis. First, in selecting explanatory variables for the multivariate Cox regression analysis, the number of major adverse cardiovascular events (MACEs) in this study (n = 74) was taken into consideration, and statistical analysis was performed to select seven or eight covariates. Specifically, 11 factors that showed differences between group H and group L, and/or depending on the presence or absence of MACEs, were adjusted each other using stepwise analysis, and eight factors (i.e., age, WBPT, diabetes, eGFR, cutaneous AF, r-AIx, albuminuria, and use of RAS inhibitors) were selected as covariates for the multivariate Cox regression analysis in this study. Cut-off values for MACEs of age, skin AF, and r-AIx are decided by receiver operating characteristic curve analysis. P values < 0.05 denoted statistically significant differences.
| Results | ▴Top |
Follow-up and grouping
The patients were followed up until June 2025. The endpoint of this study was the occurrence of primary CVD events, which were defined as MACEs. These MACEs were a composite of cardiovascular death, nonfatal ischemic heart disease, and non-fatal ischemic stroke. Patients were assigned into two groups according to the cut-off value of the WBPT, which was estimated by receiver operating characteristic curve analysis for MACEs (Fig. 1): low (group L, WBPT ≤ 74.3 s; n = 255) or high (group H, WBPT > 74.3 s; n = 162). The author’s previous report in patients with traditional cardiovascular risk factors also showed WBPT values for MACEs similar to those in this study [8].
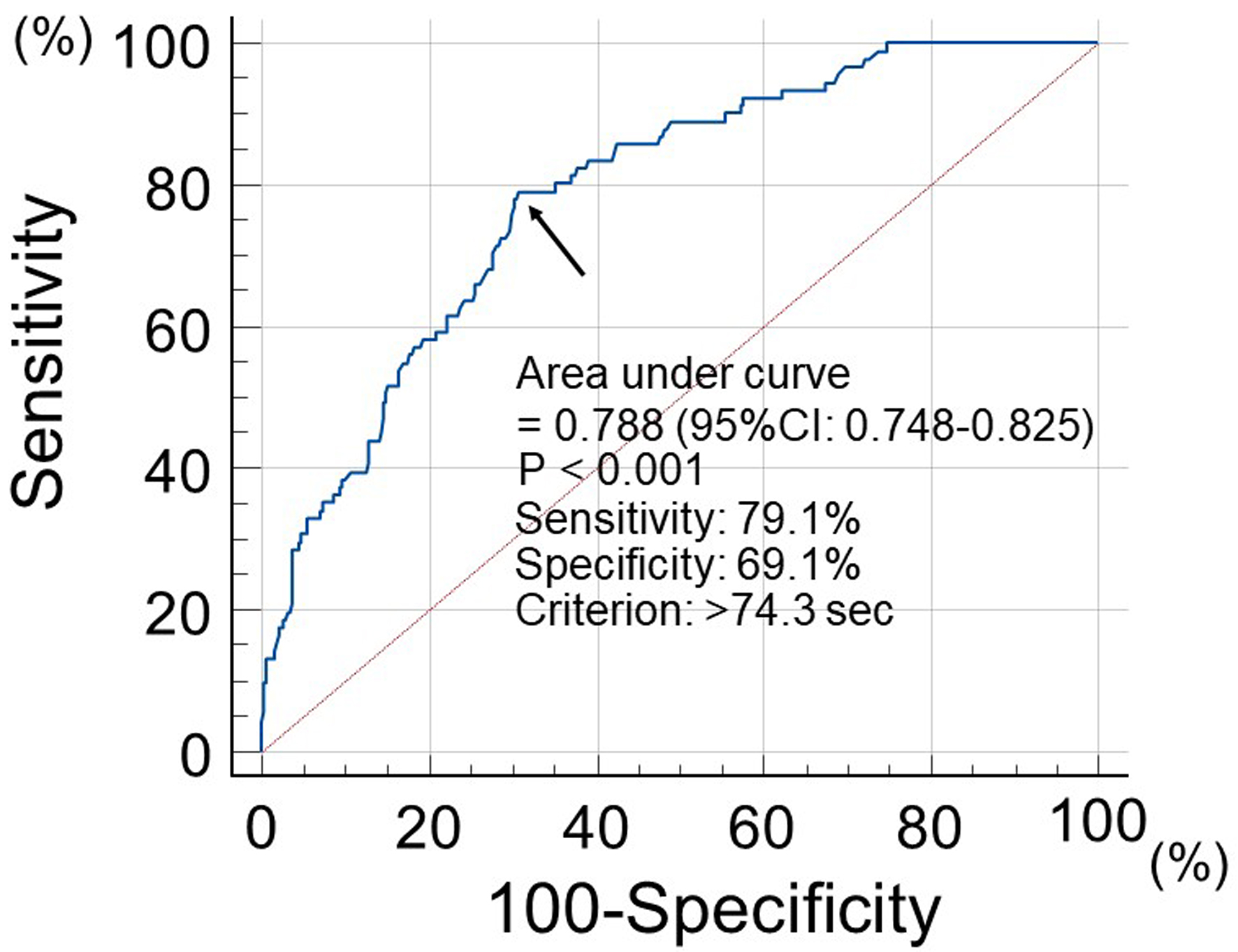 Click for large image | Figure 1. Prediction value of MACEs incidence at follow-up period using WBPT. Receiver operating characteristic curve analysis indicated that a cut-off value for WBPT of 74.3 s yielded the largest area under the curve to predict MACEs. Arrows indicate the optimal cut-off point. MACEs: major adverse cardiovascular events; WBPT: whole blood passage time. |
Clinical characteristics
Table 1 shows the clinical characteristics of the studied groups. The mean WBPT for groups L and H was 55.9 and 88.5 s, respectively. Group H was associated with a significantly higher frequency of smoking, incidence of diabetes mellitus, hematocrit, serum triglyceride levels, skin AF, incidence of albuminuria, and r-AIx compared with group L. However, group H had significantly lower eGFR, high-density cholesterol levels, and frequency of RAS inhibitor use than group L.
 Click to view | Table 1. Clinical Characteristics of the Studied Groups |
Kaplan–Meier curve analysis
Figure 2 shows the Kaplan–Meier curve for the incidence of MACEs. The median follow-up was 76 months (range: 2–96 months). During the follow-up, 74 patients developed MACEs (group L: 17 cases (6.7%)); group H: 57 cases (35.2%)). Group H had a significantly higher incidence of MACEs versus group L (log-rank test, P < 0.001).
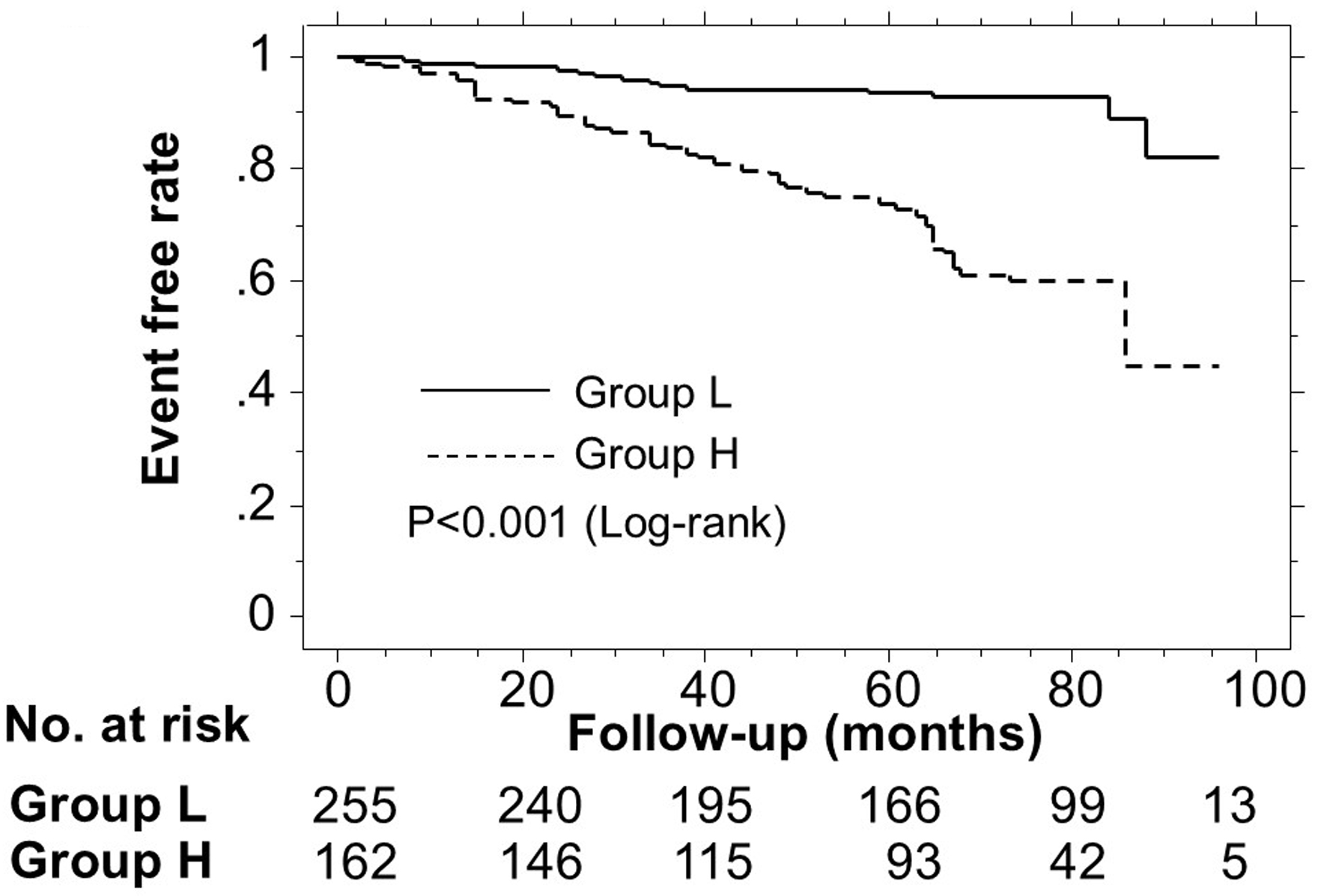 Click for large image | Figure 2. Kaplan–Meier curve analysis for the incidence of MACEs. The Kaplan–Meier curve confirmed that group H had a significantly higher incidence of MACEs compared with group L (log-rank test, P < 0.001). MACEs: major adverse cardiovascular events. |
Relationship between MACEs and clinical parameters
Table 2 presents the association between the presence or absence of MACEs and each clinical parameter. Age, incidence of diabetes mellitus, fasting blood glucose levels, skin AF, frequency of albuminuria, and r-AIx were significantly higher in patients who experienced MACEs compared with those who did not. However, patients who experienced MACEs had significantly lower eGFR and RAS inhibitor use than those who did not.
 Click to view | Table 2. Parameters at Registration of Patients With and Without MACEs |
Multivariate Cox regression analysis
Table 3 shows the results of the multivariate Cox regression analysis for MACEs. Among the eight factors, six factors (i.e., WBPT, diabetes mellitus, age, RAS inhibitor use, skin AF, and r-AIx) exhibited a significant hazard ratio for MACEs. However, the multivariate Cox regression analysis did not identify eGFR and albuminuria as significant factors for MACEs.
 Click to view | Table 3. Multivariate Cox Regression Analysis for MACEs |
| Discussion | ▴Top |
This study examined the clinical utility of blood rheology as a predictive indicator of CVD events in stage G3 CKD patients who are frequently detected in daily practice. The results of this analysis highlighted WBPT, an indicator of blood rheology, as an independent predictor of MACEs development in such patients. Therefore, it is hypothesized that patients who are at high risk of experiencing CVD events groups can be identified by measuring the WBPT as a marker of blood rheology. Similar to this study, previous studies have demonstrated the predictive value of classical CVD risk factors (aging and diabetes mellitus) for the occurrence of CVD events in CKD patients [23–25]. In addition, relatively novel CVD risk factors (skin AF and r-AIx) were also selected as independent predictors of MACEs development in this study. Furthermore, skin AF and r-AIx were significantly related to WBPT in univariate analysis. However, RAS inhibitor use was selected as a predictor to inhibit the onset of MACEs.
Significance of WBPT as a CVD risk factor in CKD
The WBPT method uses the whole blood of the patient to determine blood rheology in the artificial blood vessel. Factors of whole blood rheology include leukocyte adhesion, erythrocyte deformation, platelet aggregation, and plasma viscosity; it is speculated that WBPT is defined by these factors. However, several studies have reported that the abovementioned individual factors play a role in the onset of CVD events [26–28]. Therefore, previous studies suggest that WBPT can be a predictor of MACEs in stage G3 CKD patients. Researchers have emphasized that low eGFR and albuminuria are significant factors in the development of CVD events [29, 30]. This study revealed that these factors differed significantly between patients with and without MACEs; however, this statistically significant difference was not observed in the multivariate Cox regression analysis. The importance of low eGFR and albuminuria in the development of CVD events is obvious. In addition, the number of cases in this study was relatively small, and as a result, it is presumed that no significant differences in eGFR and albuminuria were observed. However, a statistically significant association was found between high WBPT and low eGFR and albuminuria in univariate analysis, although it is not clear as to the mechanism. Thus, the results of this study are considered that the increase in WBPT on the background of the low eGFR and the albuminuria is a more important factor for the development of MACEs in stage G3 CKD patients.
Relationships between AGEs and blood rheology
Meerwaldt et al performed skin biopsy samples collected from the same site and revealed a significant correlation between skin AF values and the levels of AGEs [31]. Therefore, skin AF is measured at a site where capillaries are located. However, the artificial blood vessel used in MC-FAN also assumes the presence of capillaries. Hence, it is presumed that skin AF represents the accumulation of AGEs in the blood vessel wall caused by the impairment of blood rheology in the capillaries. Moreover, several researchers have reported that AGEs influence blood rheology through mechanisms of abnormal blood cell function [32–34]. These results indicated an association between AGEs and blood rheology. However, some studies have shown the relationship between skin AF and the AGEs in heart and brain. Hofmann et al indicated that skin AF was significantly associated with AGEs-modified cardiac tissue collagen [35]. Also, Ohnuki et al clarified that skin AF was significantly linked to chronic cerebral infarction, including lacunar infarction, which is caused by the occlusion of a small-sized vessel [36]. Furthermore, several investigators have reported the importance of the vasa vasorum flow in arterial function or plaque vulnerability [37–39]. Hemorheological impairment in the vasa vasorum, which reflects high WBPT, may increase the risk of arterial dysfunction or plaque rupture in the heart and brain. Therefore, the significant association between skin AF and WBPT detected in this study suggested a close relationship between AGEs and impairment of blood rheology in important organs related to CVD events (heart and brain) in patients with stage G3 CKD.
Relationships between arterial reflected waves and blood rheology
Some studies on CKD have shown that AIx is associated with the presence of CVD [40, 41]. Additionally, the present investigation revealed for the first time that r-AIx is an independent predictor of MACEs in patients with stage G3 CKD. However, in the present study, the r-AIx was significantly higher in group H versus group L. Therefore, impairment of blood rheology may have caused an increase in arterial reflected waves. Previous reports have proposed microvascular dysfunction as a factor influencing the increase in arterial reflection waves [42, 43]. Nonetheless, the author has previously reported a significant association between WBPT and the renal resistive index [44]. The renal resistive index is an indicator of microvascular resistance in the kidney. Thus, an increase in renovascular resistance due to impairment of blood rheology in renal small-sized vessels may lead to an increase in arterial reflection waves; consequently, CVD events occurred.
Medications and blood rheology
Many studies indicated that RAS inhibitor use is effective in reducing the progression of CKD [45, 46]. Additionally, researchers have demonstrated that RAS inhibitor use reduces the development of CVD events in patients with CKD [47, 48]. Moreover, findings of this study indicated that RAS inhibitor use was independently associated with a reduction in the incidence of MACEs in patients with stage G3 CKD. Moreover, studies demonstrated that RAS inhibition is effective in improving the factors of blood rheology [49, 50]. However, in the present study, group L used 42% of RAS inhibitor, while group H used 31% of them (P = 0.027). Thus, the use of RAS inhibitor in patients with high WBPT and improved blood rheology may exert a further inhibitory effect on primary CVD events. Thus, intervention trials using RAS inhibitor that focus on blood rheology in stage G3 CKD patients are warranted. In recent years, the efficacy of sodium glucose cotransporter 2 (SGLT2) inhibitors in preventing the development of CVD events in patients with CKD has been demonstrated [51, 52]. However, several studies have shown that SGLT2 inhibitor has a beneficial effect on blood rheology [53, 54]. Although none of the patients used SGLT2 inhibitor at baseline, further research on the benefits of SGLT2 inhibitor in preventing the CVD events in stage G3 CKD patients is desired from the perspective of WBPT. Existing reports have shown a significant relationship between WBPT and smoking [55, 56]. In addition, Shimada et al reported that WBPT significantly decreased with smoking cessation [56]. However, in this study, no significant association was found between smoking habits and the incidence of primary CVD events, although group H had a significantly higher number of smokers compared to group L. Therefore, the results of this study suggest that smoking may contribute to the onset of CVD events in stage G3 CKD patients through a decrease in blood rheology. In recent years, the efficacy of varenicline in smoking cessation treatment has been documented [57, 58]. Therefore, for smokers with high WBPT, proactive interventional treatment including varenicline is expected to help prevent CVD through improved blood rheology in these patients.
Limitations
The limitations of this study should be acknowledged. Firstly, this analysis was performed at a single center in Japan with relatively small number. Consequently, the findings may not be generalizable to other populations. Secondly, exclusion of patients without baseline clinical data including WBPT may introduce selection bias. Thirdly, in this study, the Japanese-specific eGFR equation was used, so its application to other ethnic groups is limited, and this should be recognized. In addition, the fact that no significant differences were observed in eGFR and albuminuria in the multivariate Cox regression analysis contradicts existing literature, and this point requires careful interpretation. Finally, while WBPT appears promising, the conclusions may overstate its utility as a predictor without external validation or intervention data. In addition, WBPT was measured only once at baseline, which limits conclusions about causality and longitudinal changes. Therefore, large, multicenter prospective trials are warranted to determine whether variations in the WBPT contribute to the development of primary CVD events in patients with stage G3 CKD, including interventional trials such as RAS inhibitor use, SGLT2 inhibitor use, and smoking cessation.
Conclusions
According to the present findings, impairment of blood rheology, determined using WBPT, is predictive of primary CVD events in patients with stage G3 CKD. Further large-scale studies are warranted to confirm whether various interventions can reduce the incidence of primary CVD events with improved WBPT in patients with stage G3 CKD.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
Informed consent was obtained from all patients who participated in this study.
Author Contributions
Takashi Hitsumoto contributed to the research planning, data acquisition and analysis, and manuscript writing and editing.
Data Availability
The author declares that data supporting the findings of this study are available within the article.
Abbreviations
AF: autofluorescence; AGEs: advanced glycation end products; CKD: chronic kidney disease; CVD: cardiovascular disease; eGFR: estimated glomerular filtration rate; MACEs: major adverse cardiovascular events; MC-FAN: microchannel array flow analyzer; r-AIx: radial augmentation index; RAS: renin–angiotensin system; SGLT2: sodium glucose cotransporter 2; WBPT: whole blood passage time
| References | ▴Top |
- Kensey KR. The mechanistic relationships between hemorheological characteristics and cardiovascular disease. Curr Med Res Opin. 2003;19(7):587-596.
doi pubmed - Cowan AQ, Cho DJ, Rosenson RS. Importance of blood rheology in the pathophysiology of atherothrombosis. Cardiovasc Drugs Ther. 2012;26(4):339-348.
doi pubmed - Kikuchi Y, Sato K, Mizuguchi Y. Modified cell-flow microchannels in a single-crystal silicon substrate and flow behavior of blood cells. Microvasc Res. 1994;47(1):126-139.
doi pubmed - Kotani K, Adachi S, Osaki Y, Kurozawa Y, Araga S. Relationship between alcohol habits and hemorheology by a micro channel method in a general population. Clin Cardiol. 2008;31(10):488-491.
doi pubmed - Yoshida K, Kimura T, Aoki T, Tsunekawa K, Araki O, Shoho Y, Nara M, et al. Fasting serum insulin levels and insulin resistance are associated with blood rheology in Japanese young adults without diabetes. J Int Med Res. 2016;44(3):496-507.
doi pubmed - Hitsumoto T. Relationship between cardiovascular risk factors and hemorheology assessed by microchannel method in patients with type 2 diabetes mellitus. Diabetol Int. 2017;8(3):316-322.
doi pubmed - Hitsumoto T. Clinical impact of hemorheology on subclinical myocardial injury in patients with hypertension. J Clin Med Res. 2018;10(12):928-935.
doi pubmed - Hitsumoto T. Usefulness of the whole blood passage time as a predictor of primary cardiovascular events in patients with traditional cardiovascular risk factors. Cardiol Res. 2018;9(4):231-238.
doi pubmed - Yamagata K, Ishida K, Sairenchi T, Takahashi H, Ohba S, Shiigai T, Narita M, et al. Risk factors for chronic kidney disease in a community-based population: a 10-year follow-up study. Kidney Int. 2007;71(2):159-166.
doi pubmed - Kim KS, Park SW, Cho YW, Kim SK. Higher prevalence and progression rate of chronic kidney disease in elderly patients with type 2 diabetes mellitus. Diabetes Metab J. 2018;42(3):224-232.
doi pubmed - Liu M, Li XC, Lu L, Cao Y, Sun RR, Chen S, Zhang PY. Cardiovascular disease and its relationship with chronic kidney disease. Eur Rev Med Pharmacol Sci. 2014;18(19):2918-2926.
pubmed - Vallianou NG, Mitesh S, Gkogkou A, Geladari E. Chronic kidney disease and cardiovascular disease: Is there any relationship? Curr Cardiol Rev. 2019;15(1):55-63.
doi pubmed - Coresh J, Selvin E, Stevens LA, Manzi J, Kusek JW, Eggers P, Van Lente F, et al. Prevalence of chronic kidney disease in the United States. JAMA. 2007;298(17):2038-2047.
doi pubmed - Hill NR, Fatoba ST, Oke JL, Hirst JA, O'Callaghan CA, Lasserson DS, Hobbs FD. Global prevalence of chronic kidney disease - a systematic review and meta-analysis. PLoS One. 2016;11(7):e0158765.
doi pubmed - Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351(13):1296-1305.
doi pubmed - Imai E, Horio M, Nitta K, Yamagata K, Iseki K, Hara S, Ura N, et al. Estimation of glomerular filtration rate by the MDRD study equation modified for Japanese patients with chronic kidney disease. Clin Exp Nephrol. 2007;11(1):41-50.
doi pubmed - Kanazawa M, Yoshiike N, Osaka T, Numba Y, Zimmet P, Inoue S. Criteria and classification of obesity in Japan and Asia-Oceania. World Rev Nutr Diet. 2005;94:1-12.
doi pubmed - Meerwaldt R, Links TP, Graaff R, Hoogenberg K, Lefrandt JD, Baynes JW, Gans RO, et al. Increased accumulation of skin advanced glycation end-products precedes and correlates with clinical manifestation of diabetic neuropathy. Diabetologia. 2005;48(8):1637-1644.
doi pubmed - Hitsumoto T. Relationships between skin autofluorescence and cardio-ankle vascular index in Japanese male patients with metabolic syndrome. Cardiol Res. 2019;10(3):172-180.
doi pubmed - Kohara K, Tabara Y, Oshiumi A, Miyawaki Y, Kobayashi T, Miki T. Radial augmentation index: a useful and easily obtainable parameter for vascular aging. Am J Hypertens. 2005;18(1 Pt 2):11S-14S.
doi pubmed - Hitsumoto T. Clinical significance of the augmentation index in patients with preserved kidney function. J Nippon Med Sch. 2012;79(6):422-429.
doi pubmed - Schisterman EF, Perkins NJ, Liu A, Bondell H. Optimal cut-point and its corresponding Youden Index to discriminate individuals using pooled blood samples. Epidemiology. 2005;16(1):73-81.
doi pubmed - Boudoulas KD, Triposkiadis F, Parissis J, Butler J, Boudoulas H. The cardio-renal interrelationship. Prog Cardiovasc Dis. 2017;59(6):636-648.
doi pubmed - Hitsumoto T. Clinical usefulness of the cardio-ankle vascular index as a predictor of primary cardiovascular events in patients with chronic kidney disease. J Clin Med Res. 2018;10(12):883-890.
doi pubmed - Hahr AJ, Molitch ME. Management of diabetes mellitus in patients with CKD: core curriculum 2022. Am J Kidney Dis. 2022;79(5):728-736.
doi pubmed - Khodadi E. Platelet function in cardiovascular disease: activation of molecules and activation by molecules. Cardiovasc Toxicol. 2020;20(1):1-10.
doi pubmed - Peters SA, Woodward M, Rumley A, Tunstall-Pedoe HD, Lowe GD. Plasma and blood viscosity in the prediction of cardiovascular disease and mortality in the Scottish Heart Health Extended Cohort Study. Eur J Prev Cardiol. 2017;24(2):161-167.
doi pubmed - Tikhomirova I, Petrochenko E, Muravyov A, Malysheva Y, Petrochenko A, Yakusevich V, Oslyakova A. Microcirculation and blood rheology abnormalities in chronic heart failure. Clin Hemorheol Microcirc. 2017;65(4):383-391.
doi pubmed - Shara NM, Resnick HE, Lu L, Xu J, Vupputuri S, Howard BV, Umans JG. Decreased GFR estimated by MDRD or Cockcroft-Gault equation predicts incident CVD: the strong heart study. J Nephrol. 2009;22(3):373-380.
pubmed - Bouchi R, Babazono T, Yoshida N, Nyumura I, Toya K, Hayashi T, Hanai K, et al. Association of albuminuria and reduced estimated glomerular filtration rate with incident stroke and coronary artery disease in patients with type 2 diabetes. Hypertens Res. 2010;33(12):1298-1304.
doi pubmed - Meerwaldt R, Graaff R, Oomen PHN, Links TP, Jager JJ, Alderson NL, Thorpe SR, et al. Simple non-invasive assessment of advanced glycation endproduct accumulation. Diabetologia. 2004;47(7):1324-1330.
doi pubmed - Arriagada-Petersen C, Fernandez P, Gomez M, Ravello N, Palomo I, Fuentes E, Avila F. Effect of advanced glycation end products on platelet activation and aggregation: a comparative study of the role of glyoxal and methylglyoxal. Platelets. 2021;32(4):507-515.
doi pubmed - Morigi M, Angioletti S, Imberti B, Donadelli R, Micheletti G, Figliuzzi M, Remuzzi A, et al. Leukocyte-endothelial interaction is augmented by high glucose concentrations and hyperglycemia in a NF-kB-dependent fashion. J Clin Invest. 1998;101(9):1905-1915.
doi pubmed - Awasthi S, Gayathiri SK, Ramya R, Duraichelvan R, Dhason A, Saraswathi NT. Advanced glycation-modified human serum albumin evokes alterations in membrane and eryptosis in erythrocytes. Appl Biochem Biotechnol. 2015;177(5):1013-1024.
doi pubmed - Hofmann B, Jacobs K, Navarrete Santos A, Wienke A, Silber RE, Simm A. Relationship between cardiac tissue glycation and skin autofluorescence in patients with coronary artery disease. Diabetes Metab. 2015;41(5):410-415.
doi pubmed - Ohnuki Y, Nagano R, Takizawa S, Takagi S, Miyata T. Advanced glycation end products in patients with cerebral infarction. Intern Med. 2009;48(8):587-591.
doi pubmed - Angouras D, Sokolis DP, Dosios T, Kostomitsopoulos N, Boudoulas H, Skalkeas G, Karayannacos PE. Effect of impaired vasa vasorum flow on the structure and mechanics of the thoracic aorta: implications for the pathogenesis of aortic dissection. Eur J Cardiothorac Surg. 2000;17(4):468-473.
doi pubmed - Shimizu K, Takahashi M, Sato S, Saiki A, Nagayama D, Harada M, Miyazaki C, et al. Rapid rise of cardio-ankle vascular index may be a trigger of Cerebro-cardiovascular events: proposal of smooth muscle cell contraction theory for plaque rupture. Vasc Health Risk Manag. 2021;17:37-47.
doi pubmed - Shirai K, Hitsumoto T, Sato S, Takahashi M, Saiki A, Nagayama D, Ohira M, et al. The process of plaque rupture: the role of vasa vasorum and medial smooth muscle contraction monitored by the cardio-ankle vascular index. J Clin Med. 2023;12(23):7436.
doi pubmed - Mukai H, Svedberg O, Lindholm B, Dai L, Heimburger O, Barany P, Anderstam B, et al. Skin autofluorescence, arterial stiffness and Framingham risk score as predictors of clinical outcome in chronic kidney disease patients: a cohort study. Nephrol Dial Transplant. 2019;34(3):442-448.
doi pubmed - Covic A, Haydar AA, Bhamra-Ariza P, Gusbeth-Tatomir P, Goldsmith DJ. Aortic pulse wave velocity and arterial wave reflections predict the extent and severity of coronary artery disease in chronic kidney disease patients. J Nephrol. 2005;18(4):388-396.
pubmed - Fischer-Rasokat U, Spyridopoulos I, Walter J, Honold J, Zeiher AM, Fichtlscherer S. Microvascular dysfunction and pulse wave reflection characterize different vascular pathologies in patients at cardiovascular risk. Vasa. 2012;41(3):192-199.
doi pubmed - Rizzoni D, De Ciuceis C, Salvetti M, Paini A, Rossini C, Agabiti-Rosei C, Muiesan ML. Interactions between macro- and micro-circulation: are they relevant? High Blood Press Cardiovasc Prev. 2015;22(2):119-128.
doi pubmed - Hitsumoto T. Relationship between hemorheology assessed using microchannel array flow analyzer and kidney function in hypertensive patients. Cardiol Res. 2017;8(4):147-153.
doi pubmed - Ruggenenti P, Cravedi P, Remuzzi G. Mechanisms and treatment of CKD. J Am Soc Nephrol. 2012;23(12):1917-1928.
doi pubmed - Huang QF, Hoshide S, Cheng HM, Park S, Park CG, Chen CH, Kario K, et al. Management of hypertension in patients with chronic kidney disease in Asia. Curr Hypertens Rev. 2016;12(3):181-185.
doi pubmed - Lerma EV. Diagnosis 101: diabetic kidney disease. Clin Kidney J. 2022;15(10):1797-1799.
doi pubmed - Fu EL, Evans M, Clase CM, Tomlinson LA, van Diepen M, Dekker FW, Carrero JJ. Stopping renin-angiotensin system inhibitors in patients with advanced CKD and risk of adverse outcomes: a nationwide study. J Am Soc Nephrol. 2021;32(2):424-435.
doi pubmed - Brown NJ, Agirbasli MA, Williams GH, Litchfield WR, Vaughan DE. Effect of activation and inhibition of the renin-angiotensin system on plasma PAI-1. Hypertension. 1998;32(6):965-971.
doi pubmed - Dusing R. Pharmacological interventions into the renin-angiotensin system with ACE inhibitors and angiotensin II receptor antagonists: effects beyond blood pressure lowering. Ther Adv Cardiovasc Dis. 2016;10(3):151-161.
doi pubmed - Wheeler DC, Stefansson BV, Jongs N, Chertow GM, Greene T, Hou FF, McMurray JJV, et al. Effects of dapagliflozin on major adverse kidney and cardiovascular events in patients with diabetic and non-diabetic chronic kidney disease: a prespecified analysis from the DAPA-CKD trial. Lancet Diabetes Endocrinol. 2021;9(1):22-31.
doi pubmed - Podesta MA, Sabiu G, Galassi A, Ciceri P, Cozzolino M. SGLT2 inhibitors in diabetic and non-diabetic chronic kidney disease. Biomedicines. 2023;11(2):279.
doi pubmed - Yaribeygi H, Atkin SL, Jamialahmadi T, Sahebkar A. A review on the effects of new anti-diabetic drugs on platelet function. Endocr Metab Immune Disord Drug Targets. 2020;20(3):328-334.
doi pubmed - Ott C, Jumar A, Striepe K, Friedrich S, Karg MV, Bramlage P, Schmieder RE. A randomised study of the impact of the SGLT2 inhibitor dapagliflozin on microvascular and macrovascular circulation. Cardiovasc Diabetol. 2017;16(1):26.
doi pubmed - Hitsumoto T. Factors affecting impairment of blood rheology in obese subjects. J Cardiol. 2012;60(5):401-406.
doi pubmed - Shimada S, Hasegawa K, Wada H, Terashima S, Satoh-Asahara N, Yamakage H, Kitaoka S, et al. High blood viscosity is closely associated with cigarette smoking and markedly reduced by smoking cessation. Circ J. 2011;75(1):185-189.
doi pubmed - Jordan CJ, Xi ZX. Discovery and development of varenicline for smoking cessation. Expert Opin Drug Discov. 2018;13(7):671-683.
doi pubmed - Rahimi F, Massoudifar A, Rahimi R. Smoking cessation pharmacotherapy; varenicline or bupropion? Daru. 2024;32(2):901-906.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Clinical Medicine Research is published by Elmer Press Inc.