Point-of-Care Ultrasound to Evaluate Gastric Volumes During Gastric and Post-Pyloric Enteral Feeds in Infants and Children Undergoing Mechanical Ventilation: A Pilot Study to Assess Feasibility Study
DOI:
https://doi.org/10.14740/jocmr6506Keywords:
Point-of-care ultrasound, Enteral feedings, Aspiration, UltrasonographyAbstract
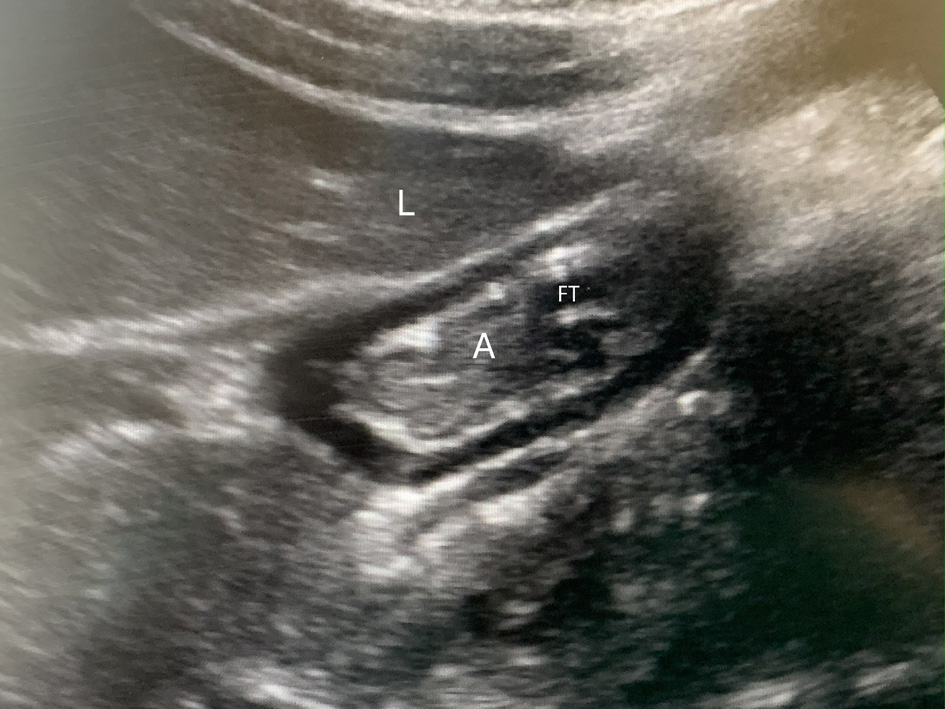
Background: In all acutely ill patients, adequate nutrition is essential to restore physiologic homeostasis and improve outcomes. Whenever feasible, the enteral route is preferred. In various settings, post-pyloric (nasoduodenal (ND) or nasojejunal (NJ)) feeds may be preferred not only to accelerate attainment of goal feeding volumes, but also to limit gastric volumes in order to decrease the potential risk of aspiration. The current study uses point-of-care ultrasound (POCUS) to evaluate gastric volume and content during enteral feedings in pediatric-aged patients receiving mechanical ventilation in the pediatric intensive care unit (ICU).
Methods: Gastric POCUS was performed to evaluate gastric contents in pediatric ICU patients, aged 0–18 years, receiving either gastric or post-pyloric enteral feedings at ≥ 50% goal. The patients were endotracheally intubated and receiving mechanical ventilation.
Results: The study cohort included 45 patients, 29 receiving nasogastric (NG) feeds and 16 receiving NJ feeds. The majority of patients (81%) receiving post-pyloric feeds had gastric volumes ≤ 0.4 mL/kg and none had volumes ≥ 2 mL/kg while more than half of NG-fed patients (53%) had gastric volumes ≥ 2 mL/kg. Only three of 18 patients (18.8%) receiving NJ feeds had a gastric volume greater than 0.4 mL/kg. When grading the aspiration risk, there was a higher aspiration risk with NG feeds compared to NJ feeds (33/39 versus 0/18, P < 0.001).
Conclusion: Gastric volumes and hence the potential aspiration risk is decreased in patients receiving post-pyloric compared to NG feeds.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.








